HP Agents and Biochemical Interactions#
Hikari A. I. Yoshihara, Ph.D.
Laboratory for Functional and Metabolic Imaging, Institute of Physics, École Polytechnique Fédérale de Lausanne (EPFL), Lausanne, Switzerland
Abstract:
The intense but transient increase in nuclear magnetic resonance signal provided by dynamic nuclear polarization (DNP) has enabled a range of new applications, including rapid, minimally-invasive metabolic imaging at the second timescale. This chapter provides an overview of the hyperpolarized carbon-13 labeled agents prepared by dissolution-DNP used in vivo and their biochemical interactions, categorized by metabolic pathway. The required characteristics of a suitable probe and an efficiently-polarizing formulation are described, as are practical considerations for formulation development and metabolite identification. Relevant biochemical principles to aid in the interpretation of the apparent metabolism of the probes are introduced with illustrative examples.
Key Words: Dynamic nuclear polarization, Carbon-13, Metabolism, Metabolic imaging, Magnetic resonance
Introduction#
The ability to directly and rapidly probe and image metabolism with high sensitivity is arguably the key feature that distinguishes hyperpolarized (HP) methods, and HP 13C-labeled agents play a critical role by providing the spatial and metabolic or spectral contrast. The metabolic sensitivity of a particular HP 13C agent depends on its particular biochemical interactions. This section provides an overview of the most commonly used HP 13C agents for dissolution DNP in pre-clinical studies, their biochemical interactions and transformations, along with some more specialized examples. The principles for their formulation for efficient polarization, and practical methods for their development are also presented.
The topics introduced in this chapter have been covered in greater depth in several recent review articles (1-5). Readers seeking more detailed information are referred to them and the primary references cited therein.
Diagram from Myriam M. Chaumeil, Chloé Najac, Sabrina M. Ronen, Chapter One - Studies of Metabolism Using 13C MRS of Hyperpolarized Probes, Editor(s): Christian M. Metallo, Methods in Enzymology, Academic Press, 561, 2015, 1-71, https://doi.org/10.1016/bs.mie.2015.04.001.
Main differences between 1H MRI and HP 13C spectroscopy and imaging#
Whereas conventional MRI generally detects the highly abundant water 1H signal, HP 13C detects only the infused agent with virtually no background signal. Water provides little chemical shift dispersion. and 1H MRI generally does not generate image contrast on the basis of chemical shift, with the exception of water-fat imaging and chemical exchange saturation transfer imaging (CEST). By contrast, most applications of HP 13C depend on the change in chemical shift resulting from metabolic transformations or the environmental conditions; however, comparatively inert HP 13C agents, such as 13C urea, can also provide contrast and physiological information from the dynamics of their distribution and their diffusion and T2 relaxation properties. The non-renewable nature of the enhanced 13C signal limits the scan time to 1-2 minutes post-infusion, as polarization is lost due to T1 relaxation and RF excitation, while 1H MRI scan times often can be extended to increase sensitivity or resolution.
Ideal characteristics of an HP 13C agent#
To provide the best sensitivity, the ideal HP 13C agent would have the highest possible polarization level and slow T1 and T2 relaxation . It would be completely biocompatible, and rapidly and specifically metabolized through the pathways of interest to provide distinct metabolite signals. In practice, it is not possible to fulfill all these requirements, but some agents come closer to meeting them than others. While one of the great advantages of HP 13C is that the metabolic probes can be the native substrates, the physical properties of the metabolites along with their biochemical interactions limit the duration and intensities of the available signals. For these reasons, some metabolic pathways – and sites of labeling – are more suitable than others for probing with HP 13C agents.
Carbon-13 magnetic resonance and labeled metabolic substrates#
Carbon-13 has a natural abundance of around 1.1% and a gyromagnetic ratio 0.2515 times 1H’s, corresponding to a sensitivity 5800-fold lower than 1H. Even when hyperpolarized, enrichment of the 13C isotope is required for the highest sensitivity. 13C agents are synthesized from 13C-enriched precursors derived from carbon monoxide distilled to separate the 13CO from 12CO. Not every carbon atom in a molecule is well suited for HP applications, and the best sites for labeling by 13C will have the following properties:
1) Slow longitudinal relaxation: Ideally, the label will have a long-lived enhanced signal in solution. This entails a long T1, as found in quaternary carbons, which lack any directly bonded hydrogens, minimizing dipolar cross-relaxation. For naturally-occurring metabolites, carboxylic acids are the main site of labeling, but ketone, acetal, amide, urea and guanidine functional groups are also labeled. The T1 of 13Cs with directly-bound hydrogen can be substantially increased by substitution with deuterium and has been used extensively as described later, although 13C–2H scalar coupling interactions (typically 20 – 25 Hz) result in a broader resonance. Carbon atoms with minimally-restricted molecular rotation, such as those in smaller molecules and the ends of chains have short rotational correlation times, which results in slower longitudinal relaxation.
2) Metabolic products with slow longitudinal relaxation [and]{.ul} a change in chemical shift: A change in the resonant frequency of the 13C label is needed between the original 13C substrate and resulting metabolites in order to probe biochemical processes. With the spectral resolution typically achievable in clinical and pre-clinical scanners, a change in chemical shift of greater than 2 ppm may be needed to reliably detect the metabolite. Fortunately, due to the large chemical shift dispersion of 13C, in most cases the resonant frequency of the labeled site is sufficiently changed by metabolism and can be easily distinguished.
Overview of biological HP agents#
Because of the requirement for slowly-relaxing 13C spin labels, hyperpolarized methods are only applicable to a subset of the metabolome. In practice, its uses are limited to small-molecule substrates labeled at privileged sites capable of sustaining hyperpolarized signals. Additionally, the rate of their metabolic conversion , discussed further in section 6.4, must be sufficiently high to provide a detectable signal from downstream products. Given these constraints, HP 13C has been particularly well-suited to applications involving aspects of energy metabolism, but some other pathways have sufficiently-high flux and appropriate substrates. This section summarizes the HP 13C agents used in pre-clinical applications, grouped according to the metabolic processes they probe.
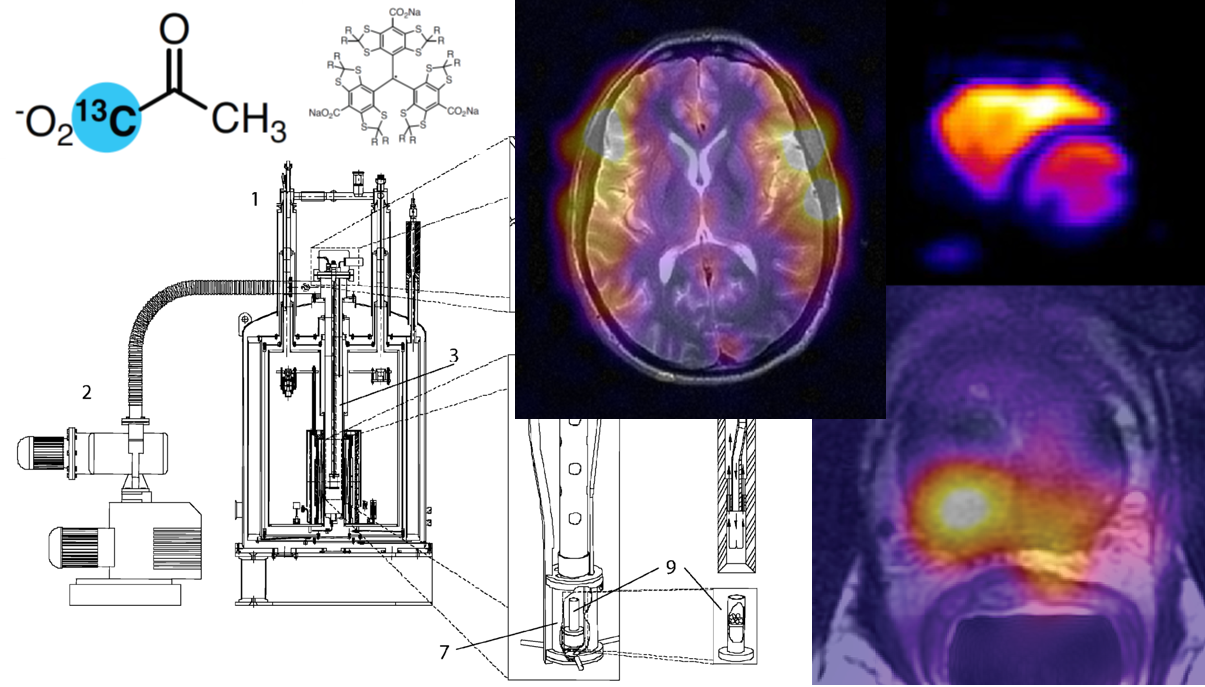
[1-13C]pyruvate#
[1-13C]pyruvate is the archetypal HP 13C agent. With a host of applications, it has excellent performance, thanks to its high polarization, rapid metabolism, and low endogenous levels. While the readily-detected conversion to [1-13C]lactate in tumors has been its main application, pyruvate lies at the junction of several important metabolic pathways. Its transamination to [1-13C]alanine is also apparent in many tissues, most prominently in the liver, and pyruvate oxidation by pyruvate dehydrogenase (PDH) liberates labeled 13CO2, readily detected by its rapid conversion to bicarbonate ion H13CO3-, particularly in organs with intense oxidative metabolism, like the heart and brain. (Figure 1). As discussed in below in section 6.2.4, [2-13C]pyruvate is also used to label metabolites downstream of PDH.
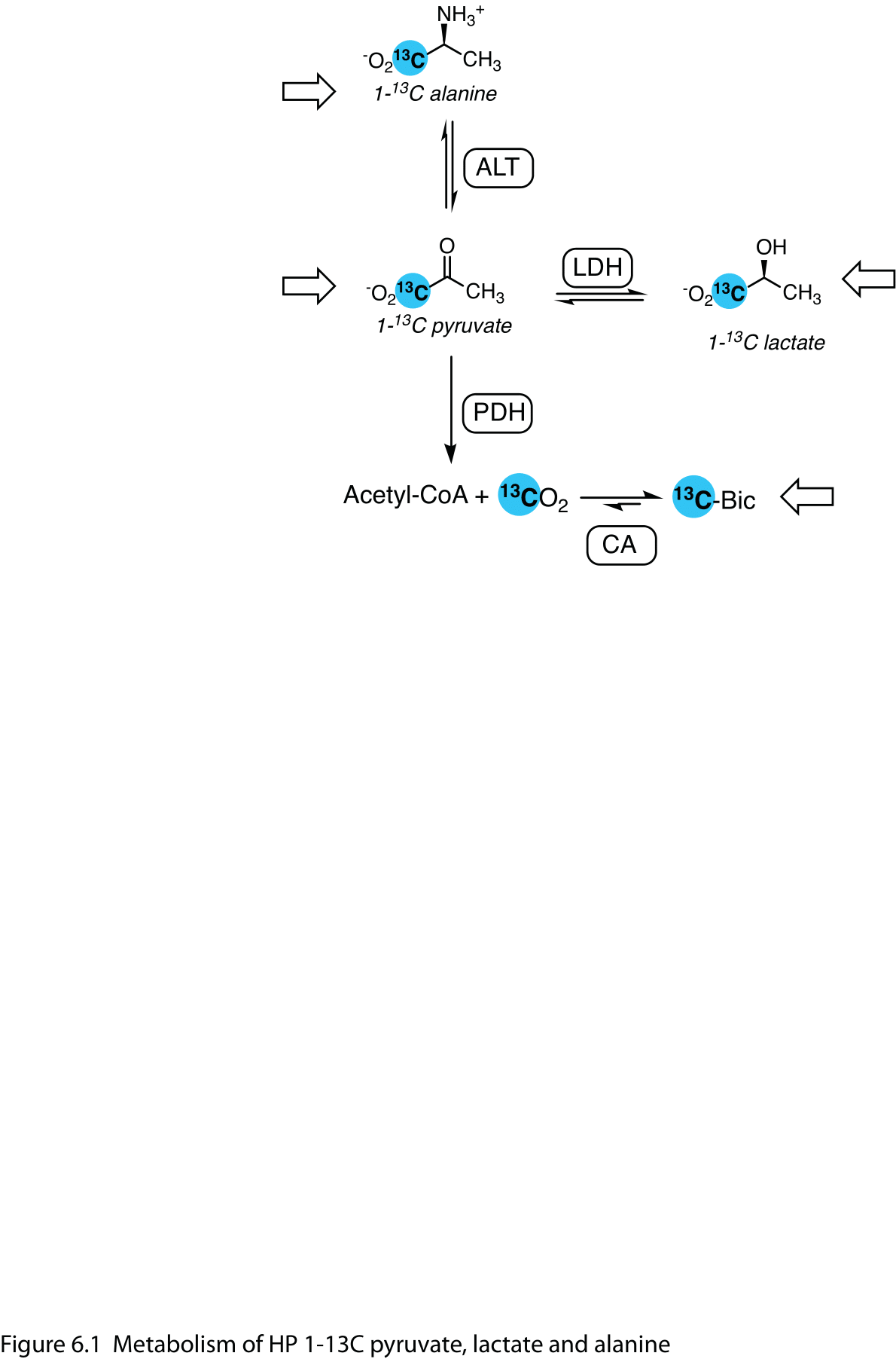
Figure 1 Metabolism of HP 1-13C pyruvate, lactate and alanine. Pyruvate, lactate and alanine are reversibly interconverted in the cell by lactate dehydrogenase (LDH) and alanine transaminase (ALT). Oxidation of pyruvate by mitochondrial pyruvate dehydrogenase (PDH) yields labeled carbon dioxide, which carbonic anhydrase (CA) rapidly interconverts with bicarbonate. Open arrows denote infused HP substrates.
Pyruvate carboxylation to oxaloacetate is an important anapleurotic pathway that supports the TCA cycle in oxidative energy metabolism. It is a first step for glucose synthesis in the liver and kidney, where metabolites downstream of pyruvate carboxylase including aspartate, malate and fumarate are produced from [1-13C]pyruvate. Of these, [1-13C]aspartate provides the most reliable signal, and it is modulated by the nutritional state (6,7). By label scrambling (Figure 6.2) via fumarate, [1-13C]oxaloacetate is converted to [4-13C]oxaloacetate, whose conversion to phosphoenolpyruvate (PEP) and entry into the gluconeogenesis pathway liberates 13CO2, detected as bicarbonate. Pyruvate-to-bicarbonate conversion in gluconeogenic tissues results from both pyruvate oxidation and PEP production (8).
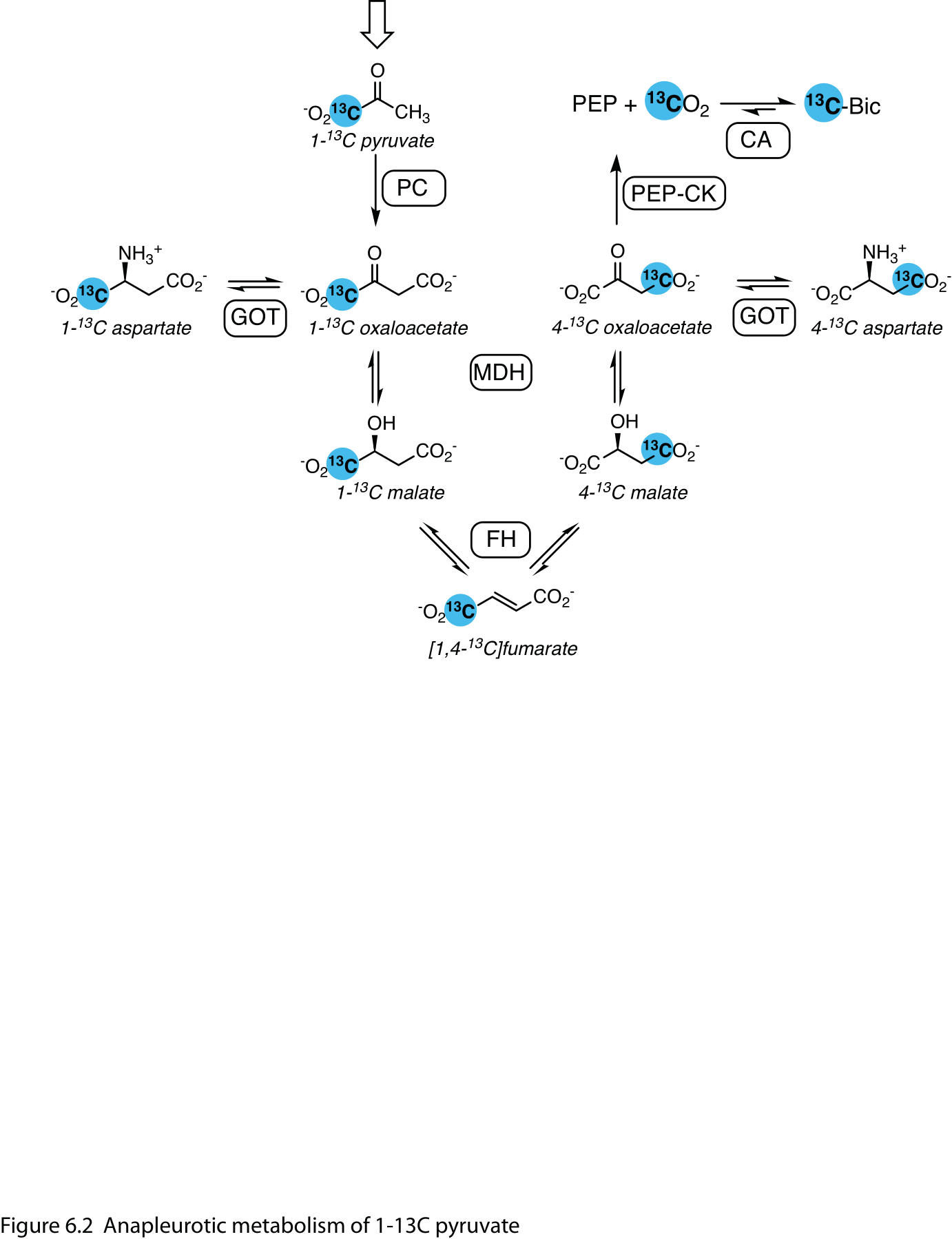
Figure 2 Anapleurotic metabolism of 1-13C pyruvate. In the mitochondrion, pyruvate is converted to the tricarboxylic (TCA) cycle intermediate oxaloacetate by pyruvate carboxylase (PC). Labels at the 1- and 4-positions are interconverted via symmetric fumarate. Aspartate is the main metabolite observed downstream of PC and the label at the 4-position is converted to bicarbonate by phosphoenolpyruvate carboxykinase (PEPCK) in gluconeogenic organs such as the liver. Abbreviations: GOT – glutamic oxaloacetic transaminase; MDH – malate dehydrogenase; CA – carbonic anhydrase.
[1,4-13C2]Fumarate#
HP [1,4-13C2]fumarate is a probe of necrotic tissue. The hallmark increase in cell membrane permeability associated with necrosis results in the increased accessibility of HP fumarate to fumarate hydratase, resulting in a marked increase in its conversion to [1,4-13C2]malate (Figure 3). This effect has been noted in treated tumors (9-11) as well as in the injured kidney (12,13) and myocardium (14).
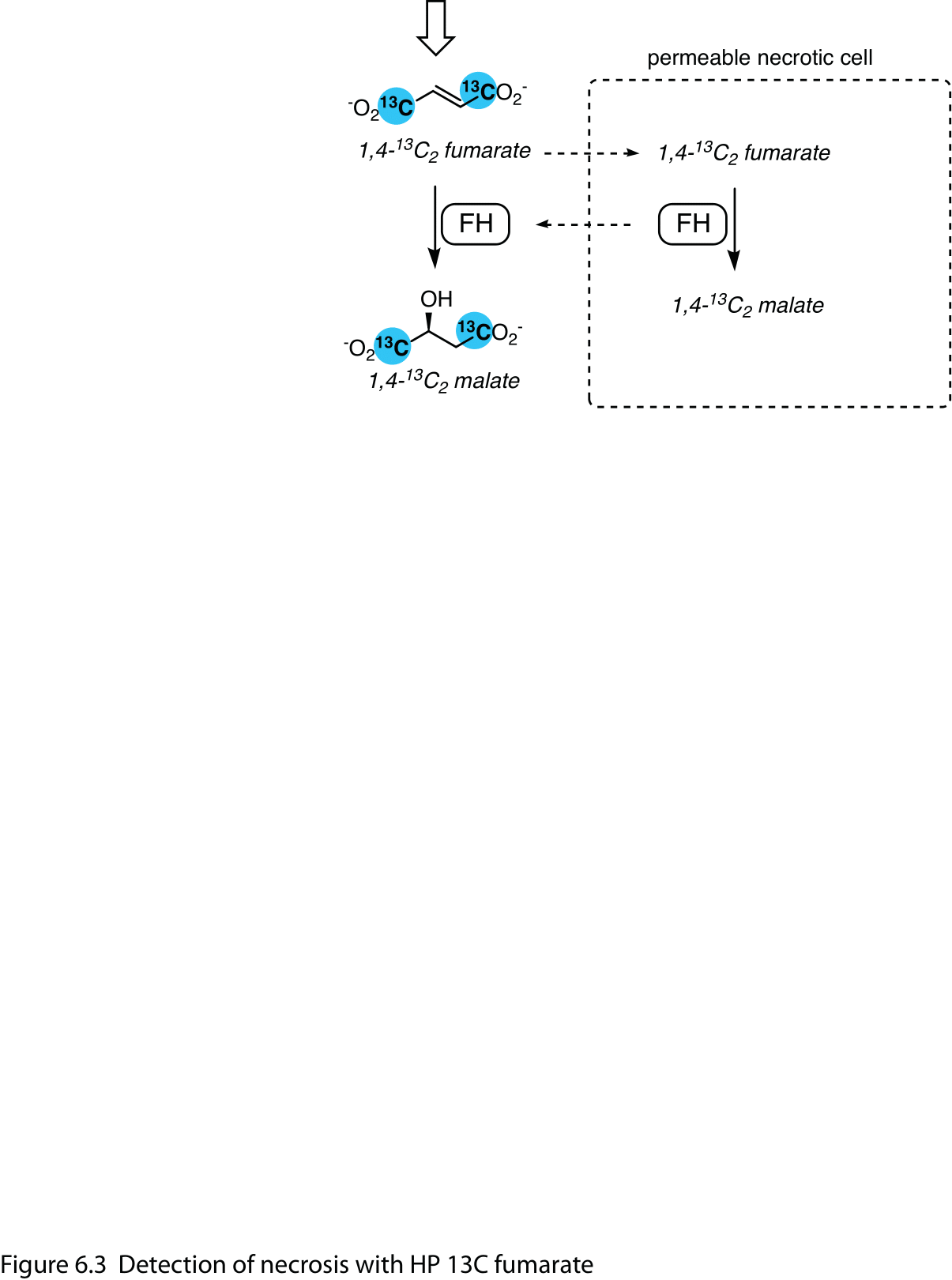
Figure 3 Detection of necrosis with HP 13C fumarate. Cell membrane disruption in necrotic tissue increases the accessibility of HP 1,4-13C2 fumarate to fumarate hydratase (FH), which efficiently converts fumarate to 1,4-13C2 malate.
Carbohydrates#
The significant metabolic roles of carbohydrates in normal metabolism and tumors make them important targets for HP 13C agents. Despite their metabolic importance, sugars are generally not ideal substrates, mainly due to the relatively short T1s of their carbon atoms. The quaternary C2 in fructose, however, has a T1 as long as 16 s at 11.7 T (15), and deuteration of the carbon-bonded hydrogens in 13C-labeled glucose slows their relaxation so that C2 – C5 have T1s in the same range (16).
The uptake and phosphorylation of HP [2-13C]fructose has been detected in mouse prostate tumors (15), as has the conversion of HP [U-13C6, 2H7]glucose to lactate, dihydroxyacetone phosphate, 6-phosphoglycerate (6PG) and bicarbonate in the EL4 lymphoma xenograft model (17). The conversion of both HP [U-13C6, 2H7]glucose and [3,4-13C2, 2,3,4,6,6-2H5]-glucose to lactate, 3-phosphoglycerate (3PG) and pyruvate is apparent in the rat brain (18), reflecting its high glucose consumption. 3-Phospho[1-13C]glycerate is an intermediate in glycolysis while 6-phospho[1-13C]gluconate is produced in the pentose phosphate pathway (PPP) (Figure 4), but there is only a small difference in chemical shift between the two, and 3,4-13C2-glucose labels only 3PG. The metabolism of glucose to 6PG in EL4 cells has been confirmed with additional experiments (19). The PPP can be more specifically probed with HP δ‐[1‐13C]gluconolactone (20) and the conversion of labeled 6PG to bicarbonate.
Smaller carbohydrates are potentially better HP 13C agents. Dihydroxy[2-13C]acetone (DHAc) is a precursor to dihydroxyacetone phosphate (DHAP), an intermediate in the middle of both the glycolysis and gluconeogenesis pathways (Figure 4). In the kidney and liver (21,22) it labels both upstream and downstream metabolites, including phosphenolpyruvate, glyceraldehyde-3-phosphate and glucose, whose relative intensities vary with the nutritional state. In vivo applications for DHAc are somewhat challenging, with the requirement of high-bandwidth acquisition methods (23) and 1H decoupling (24) to achieve the best sensitivity and resolution. [1-13C]Glycerate (25) may similarly probe these pathways, although the primary metabolites reported are lactate, pyruvate and bicarbonate.
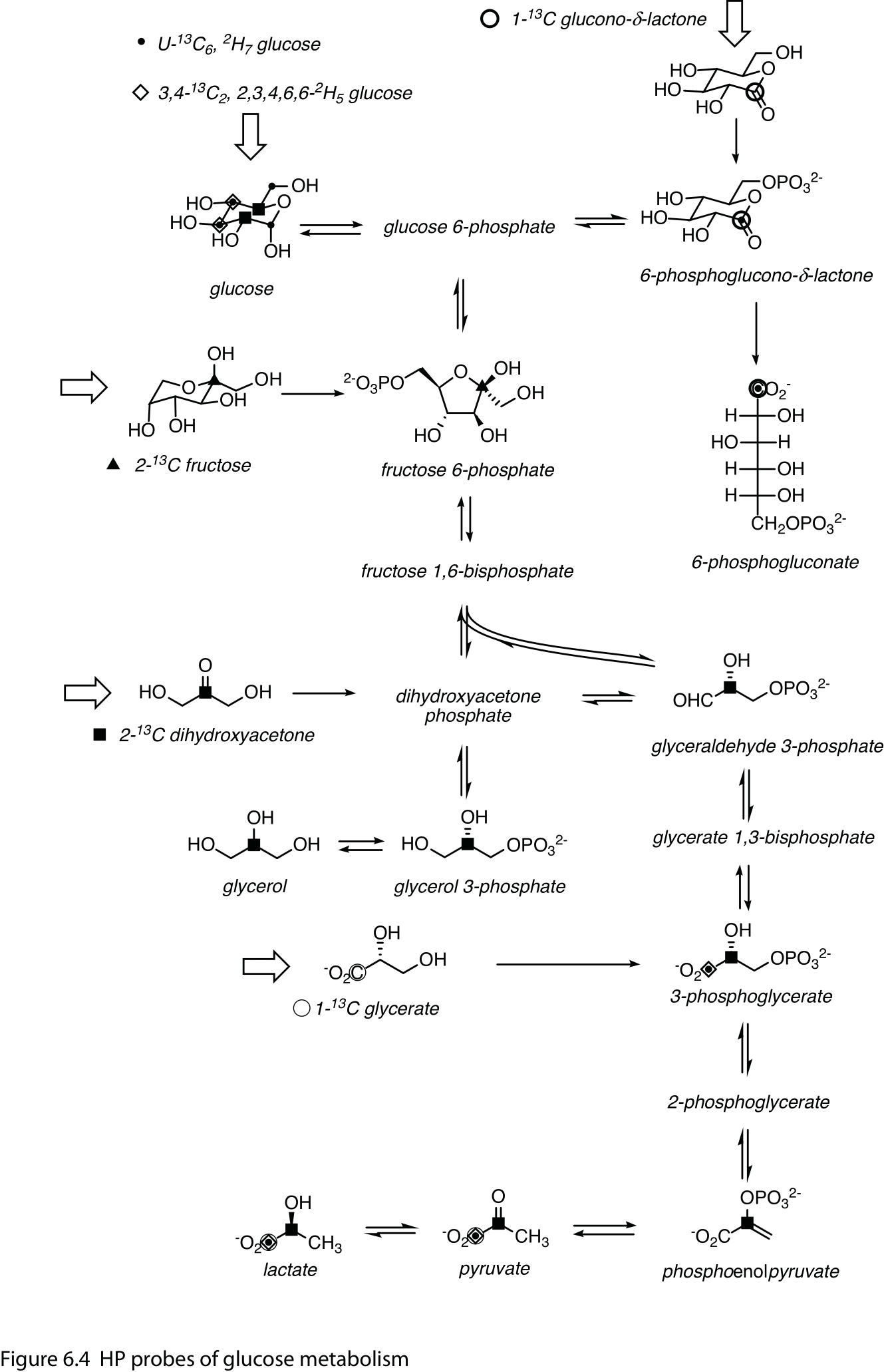
Figure 4 HP probes of glucose metabolism. Composite of HP 13C agents engaging the glycolysis, gluconeogenesis and pentose phosphate pathways. Sites of 13C-labeling for the probes and detected metabolites are indicated with the following symbols (undetected labeled metabolites/sites have been omitted for clarity): small dots – U-13C6, 2H7 glucose; open diamonds –3,4-13C2, 2,3,4,6,6-2H5 D-glucose; bold circle – δ‐1‐13C gluconolactone; triangle – 2-13C fructose; square – 2-13C dihydroxyacetone; thin circle – 1-13C glycerate. Open arrows denote infused HP substrates. Double arrows indicate transformation by different energetically-favorable reactions in the forward and reverse directions.
Fatty acids and ketone bodies#
Fatty acids are important energy substrates in several major organs and tissues, as are ketone bodies, particularly during starvation conditions. They are converted into acetyl-CoA units and oxidized further in the TCA cycle. The metabolism of short- and medium-chain fatty acids and ketone bodies labeled with HP 13C has been detected in several organs, but the heart has been the primary target organ for these studies.
Acetyl-CoA has low steady-state concentrations and provides only a faint signal, if any, in HP experiments, and the most prominent metabolites downstream of [1-13C]acetyl-CoA are [5-13C]glutamate, [5-13C]citrate, and [1-13C]acetylcarnitine (Figure 5).
[1-13C]Acetate, the simplest fatty acid, is readily converted to acetylcarnitine in the liver, skeletal muscle, heart, (26,27), and kidney (28). In the heart, its conversion to citrate can also be resolved, enabling estimation of the TCA cycle flux (29). In the brain, the primary metabolite detected is α-keto[5-13C]glutarate (30).
The myocardial metabolism of the 4- and 8-carbon fatty acids [1-13C]butyrate (31-34) and [1-13C]octanoate (35) requires β-oxidation to generate the [1-13C]acetyl-CoA intermediate, and they resonate at a higher chemical shift than acetate, allowing the [5-13C]glutamate signal to be resolved.
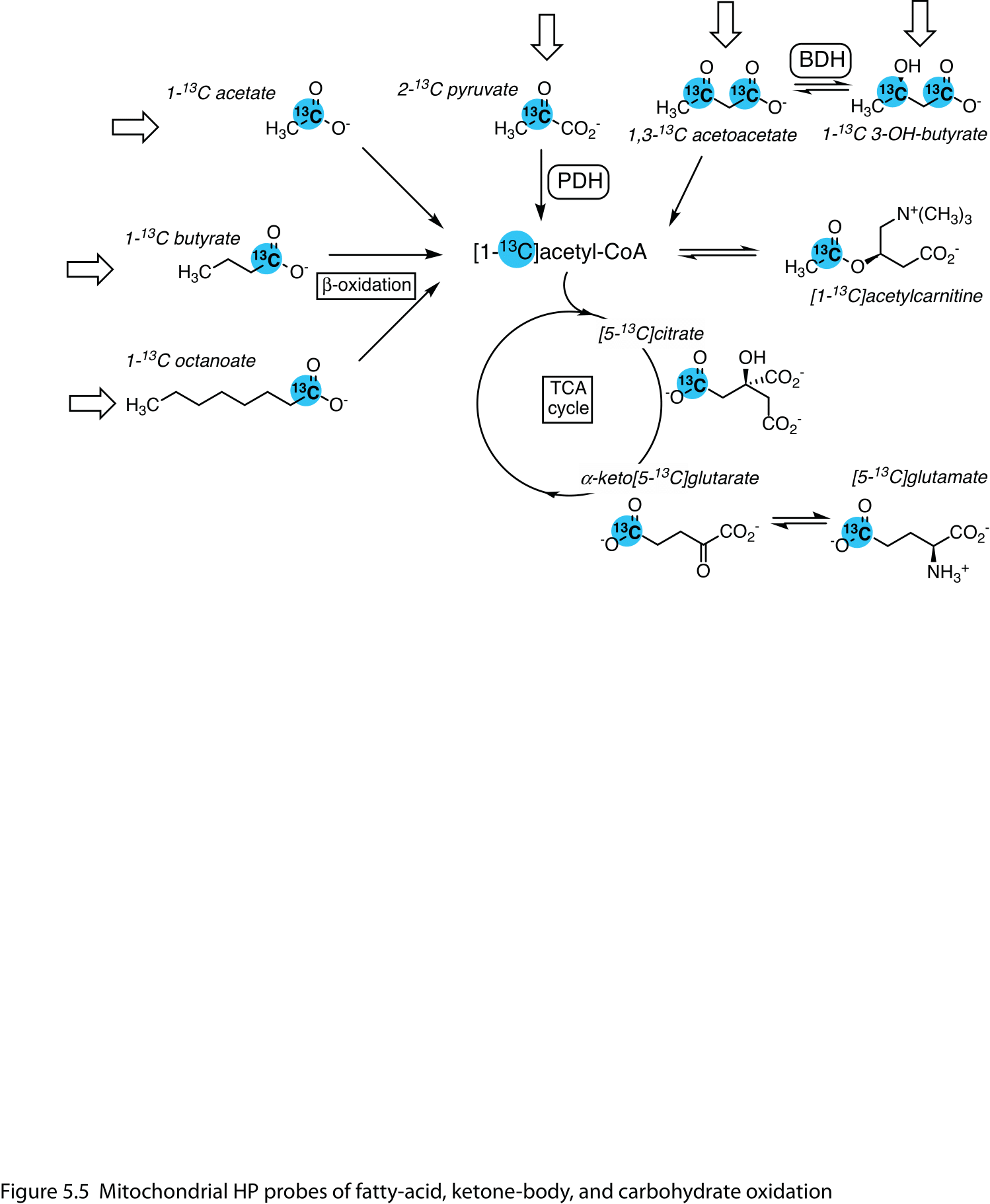
Figure 5 Mitochondrial HP probes of fatty-acid, ketone-body, and carbohydrate oxidation. They are metabolized by different routes to the common intermediate, [1-13C]acetyl-CoA, which exchanges with the acetylcarnitine pool and enters the tricarboxylic acid (TCA) cycle. Detected metabolites included [5-13C]citrate, [5-13C]glutamate and α-keto[5-13C]glutarate. Open arrows denote infused HP substrates. Abbreviations: PDH – pyruvate dehydrogenase; BDH – 3-hydroxybutyrate dehydrogenase.
The ketone bodies acetoacetate and [d]{.smallcaps}-3-hydroxybutyrate are also amenable as HP 13C agents. The myocardial metabolism of [1-13C]-, or [3-13C]acetoacetate is changed by nutritional (36), diet (34) and disease state (37), as well as treatment with an SGLT2 inhibitor (38). The reduction of [1,3-13C2]acetoacetate to [d]{.smallcaps}-3-hydroxybutyrate has also been studied in U87 brain tumors (39) and rat kidney (40) and related to changes in the NAD+ / NADH redox state.
L-[1-13C]lactate and amino acids#
With their direct conversion to pyruvate, [l]{.smallcaps}-[1-13C]alanine and [l]{.smallcaps}-[1-13C]lactate probe the same pathways (Figure 1); however, the information they provide is not precisely the same, and this can be advantageous in some situations.
HP lactate has been used as an alternative to pyruvate, where its metabolism has been detected in the liver and kidney (41-43), heart (44), skeletal muscle (45) and brain (46). Unlike pyruvate, HP lactate can readily be used at physiological concentrations, and the metabolism measured therefore better reflects its normal metabolism. However, it has lower sensitivity than pyruvate as the endogenous lactate by competes with the labeled lactate for entry into the tissue and cells. Additionally, the lactate within the cell similarly dilutes the label, resulting lower intracellular fractional enrichments and proportionally lower metabolite signals.
The liver is the main site for uptake of infused HP alanine where it is a major substrate for gluconeogenesis (47,48). A notable application of alanine is the measurement of the intracellular redox state and NAD+ / NADH ratio from the pyruvate / lactate ratio (49). Recently, the mirror-image stereoisomer [d]{.smallcaps}-[1-13C]alanine was reported as a probe of [d]{.smallcaps}-amino acid oxidase activity in the kidney and blood leukocytes (50). Compared to HP [l]{.smallcaps}-alanine, it yields the same metabolites but uses a different route.
The metabolism of other HP 13C amino acids has been detected in vivo. Glutamine is of particular interest due to the enhanced glutaminolysis seen in tumor cells (51), and increased conversion of HP [5-13C]glutamine to [5-13C]glutamate has been noted in animals with liver tumors (52,53). Glutamate is transaminated to α-ketoglutarate, which is in turn converted to 2-hydroxyglutarate (2-HG) by tumor cells that express gain-of-function mutations in isocitrate dehydrogenase-1. 2-HG production has been measured in animal models with tumors bearing this mutation, using HP α-keto[1-13C]glutarate (54) or [1-13C]glutamine (55) as the precursor. (Figure 6)
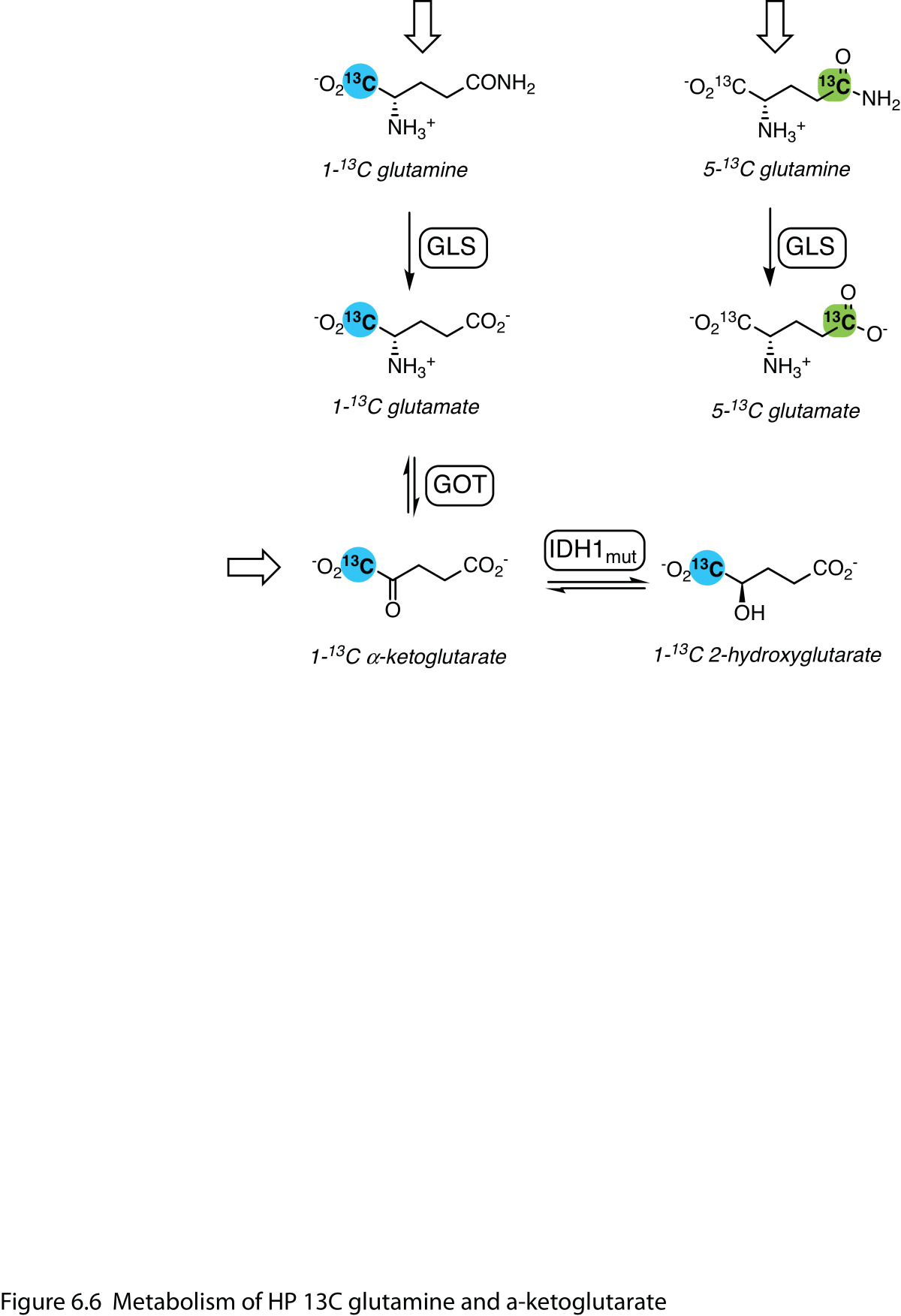
Figure 6 Metabolism of HP 13C glutamine and α-ketoglutarate. Elevated glutaminase (GLS) activity in tumors is directly detected with HP 5-13C glutamine. HP 1-13C α-ketoglutarate is converted to glutamate and is also reduced to 2-hydroxyglutarate (2-HG) in tumor cells with gain-of-function mutations in isocitrate dehydrogenase-1 (IDH1mut). HP 1-13C glutamine can also be converted to 2-HG. Open arrows denote infused HP substrates.
Peptidase activity is also detectable in vivo with HP 13C probes. The cell-surface enzyme γ-glutamyltransferase (GGT) plays an important role in glutathione metabolism. GGT is most abundantly expressed in the renal proximal tubules, and approximately 10% of HP γ-glutamyl[1-13C]glycine is converted to [1-13C]glycine in the kidney, reflecting renal function (56). Elevated GGT expression is also a biomarker of some tumors, and it is implicated in helping malignant cells respond to oxidative stress. Recently, elevated GGT activity was imaged in U87 glioblastoma tumors with HP γ-glutamyl[1-13C]glycine (57).
Redox sensors#
The redox state, the balance between reducing and oxidizing species, is an important cellular parameter that influences numerous metabolic processes and is of particular interest in metabolic imaging of cancer. The metabolism of several HP 13C agents is linked, directly or indirectly, to the redox state in the cell or in specific compartments.
The interconversion of pyruvate and lactate by lactate dehydrogenase is influenced by the redox state, and the NAD+ / NADH ratio has been estimated from the ratio of pyruvate and lactate produced from HP [1-13C]alanine in the liver (49) and from HP [U-13C6, 2H7]glucose in cultured cells (58). Similarly, the reduction of acetoacetate to [d]{.smallcaps}-3-hydroxybutyrate by 3-hydroxybutyrate dehydrogenase depends on the mitochondrial NAD+ / NADH ratio, and differences in the conversion of HP [1,3-13C2]acetoacetate has been related to changes in the redox state in brain tumors (39) and the kidney (40) .
Another redox-dependent HP probe is [1-13C]dehydroascorbate (DHA), the oxidized form of vitamin C. DHA is readily taken up into cells via glucose transporters and its reduction to vitamin C depends on glutathione or NADPH (59,60). Higher conversion of HP 13C DHA to vitamin C has been observed in tumors, and depletion of glutathione results in lower conversion (61,62).
Sensors of pH and other ions#
While most HP 13C agents are isotopologues of endogenous metabolites and probe specific enzyme-catalyzed metabolic processes, a variety of probes responsive to reactive metabolites or ions have been proposed. These include compounds that react with oxidizers such as hydrogen peroxide (63,64) and hypochloric acid (65) or bind to metal ions such as Ca2+ (66), Zn2+ (67) or Mg2+ (68).
Bicarbonate (H13CO3-) is the main pH-sensing HP 13C agent used in vivo. Using the Henderson-Hasselbalch equation, the ratio of 13CO2 and H13CO3- signals provides a readout of pH, via the rapid equilibration of carbon dioxide and carbonic acid (pKa = 6.17) by carbonic anhydrase. Infused HP 13C-bicarbonate has been used to image tissue pH (69,70), and its metabolic production from 1-13C pyruvate and PDH in the heart is sufficient to allow spectroscopic measurement (71) and imaging of myocardial pH (72). Sodium bicarbonate has relatively poor solubility, and better polarizing precursors have also been used (69,73,74) though they require additional processing prior to infusion.
Besides bicarbonate, a variety of other HP 13C pH-sensing agents have been described. They are variously based on amino-acid derivatives (66,75), buffers (76), malonate derivatives (77), and zymonic acid derived from pyruvate (78), which has also been used in vivo.
Urea and other perfusion tracers#
Some HP 13C agents are not appreciably metabolized, but this property is useful for perfusion markers. Foremost among these is urea, which is produced in the liver, eliminated by the kidneys, and otherwise remains mainly in the vascular compartment. Compounds engineered for high HP 13C performance also used as perfusion markers. Two examples are HP001 (bis-1,1-(hydroxymethyl)-[1-13C]cyclopropane-d8) (79-81) and deuterated 13C tert-butanol (2-methyl-[2-13C]propan-2-ol-d9) (82,83). Both have quaternary 13Cs with the neighboring carbon atoms deuterated to minimize dipolar cross-relaxation and hydroxyl groups to confer water solubility. The more hydrophobic tert-butanol also leaves the vascular compartment and readily crosses the blood-brain barrier (82,84).
As the original HP 13C contrast agent (85,86), 13C-urea has been used extensively as a perfusion marker to provide a measure of the delivery of co-polarized pyruvate. Renal function can be probed by imaging the dynamics of urea handling in the kidney, and differences are apparent with diuresis / antiduresis (87) and diabetes (88). Additionally, the much slower transverse relaxation of [13C,15N2]urea enables high-resolution T2-mapping, which distinguishes multiple different compartments in the kidney (89,90).
Formulation of HP 13C agents for dissolution DNP#
The need for formulation#
Formulation plays a critical role in achieving the highest attainable level of DNP enhancement for 13C-labeled compounds and is therefore a key factor in the method. There is unfortunately no single ‘magic formula’ suitable for all compounds, and a formulation optimal for polarization at one magnetic field strength and temperature may not be as well suited under other conditions. While a formulation is best optimized empirically, there are a number of general principles that can be used to guide its development.
Essential characteristics of a formulation#
The requirements for a well performing formulation are an appropriate radical, a high concentration of labeled compound and formation of an amorphous glass upon rapid cooling. DNP requires a paramagnetic species as a source of spin order. This is usually provided by a stable radical present at concentrations of 10 – 60 mM. Diffusion of spin polarization between nuclei is promoted by their proximity, and higher concentrations of 13C nuclei facilitate higher rates of polarization. (See Physics of dDNP for additional information.)
Free radicals#
A chemically-stable (“persistent”) free radical usually provides the spin order for dissolution DNP; however, other paramagnetic species, such as free radicals generated by irradiation can also be used. Free radicals used for DNP have an unpaired valence electron, resulting in net electron spin. These highly reactive species are stabilized by nearby sterically bulky substituents that prevent their quenching. Like 1H and 13C nuclei, electrons have a spin quantum number of ½, but their gyromagnetic ratio is 2617-fold higher than 13C, such that they are almost completely polarized at the temperatures and magnetic fields used for dissolution DNP and resonate at microwave frequency. Electron paramagnetic resonance (EPR) spectroscopy, which is analogous to NMR spectroscopy, is used to characterize radicals and other paramagnetic species, and as described in Physics of dDNP the mechanisms of DNP depend on the irradiation of this resonance with microwaves. Substituted triphenylmethyl (“trityl”) radicals are most commonly used, but other persistent radicals, including nitroxyl radicals such as TEMPO and BDPA (1,3-bis(diphenylene)-2-phenylallyl) are also used in some cases.
The main considerations in selecting one radical over another are: 1) its EPR line shape, 2) its solubility in aqueous or organic solution, and 3) its chemical stability As the DNP process is most efficient when the EPR line width matches the nuclear Larmor frequency (91), direct polarization of 13C with trityl radical or BDPA has been used to attain the highest signal enhancements. Depending on the compound to be polarized, the formulation may be in organic or aqueous solution and the radical must be soluble. The trityl radicals AH111501 and OX063 (Figure 6.7) are soluble in a variety of conditions and are widely applicable, whereas the “Finland” trityl radical, TEMPO and BDPA work best with organic solvents. BDPA in particular has a limited range of solubility (92). 4-oxo- and 4-hydroxy-TEMPO are good substitutes for TEMPO in aqueous formulations.
The radical must be depleted prior to infusion in clinical applications, and its presence is otherwise disadvantageous after dissolution as it can accelerate paramagnetic relaxation of the polarized spins, particularly at the low magnetic fields typically encountered during transfer from the polarizer magnet to the scanner magnet. A number of alternative approaches that yield radical-free solutions have been demonstrated but have not entered routine use. They can be generally categorized as using either immobilized radicals or quenchable radicals. Radicals immobilized in a gel matrix have been used as polarizing agents for both flow (93,94) and dissolution DNP (83) as have conjugates to mesoporous hybrid silica (95). Quenching of nitroxyl radicals with sodium ascorbate has been achieved both during dissolution (96) and after transfer (97), resulting in slower relaxation rates.
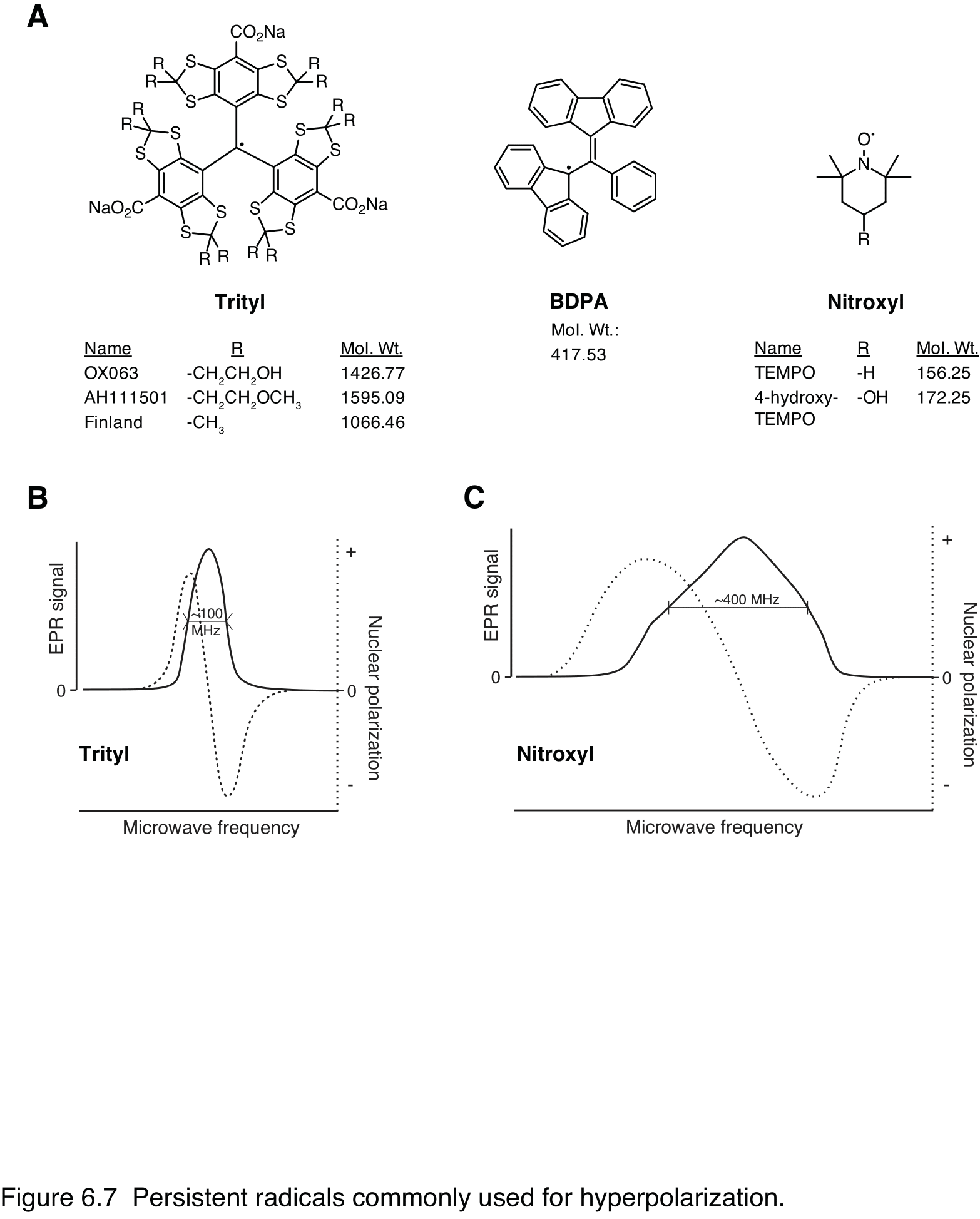
Figure 7 Persistent radicals commonly used for hyperpolarization. A: Chemical structures; dots denote the unpaired radical electrons. Trityls and BDPA are typical narrow-line radicals well suited for polarizing low-gamma nuclei such as 13C. Nitroxyls have a broader EPR resonance and can also polarize high-gamma nuclei like 1H and 19F. B: & C: Schematic plots of trityl and nitroxyl EPR spectra and solid-state DNP enhancement as a function of microwave frequency (197 GHz) at 7T and 1K. Solid trace shows absorption-mode EPR signal, with the full-width half-maximum indicated. Dotted line shows DNP enhancement by thermal mixing with positive enhancement at frequencies below the center of the electron resonance and negative enhancement at higher frequencies.
The light sensitivity of pyruvic acid and other α-keto acids can be exploited to provide an alternative to persistent radicals. Irradiation of frozen pyruvic acid with medium- or long wave ultraviolet light produces free radicals, which persist at liquid-nitrogen temperature (98) and they are suitable paramagnetic agents for DNP (99,100). The radicals are quenched upon dissolution and also by transient elevation of the temperature above ~190K, resulting in radical-free HP solid samples that relax very slowly at liquid-helium temperature (101). This enables the appealing possibility of storing or transporting HP agents to sites without a polarizer. Transient radicals prepared by UV irradiation of α-keto acids have also been used to polarize other 13C agents (33,102,103).
Glassing#
For optimal polarization, the radical needs to be evenly distributed throughout the sample volume. This usually achieved by rapid cooling of the sample solution in liquid nitrogen or liquid helium such that it freezes as an amorphous glassy solid before crystallization or solute precipitation occurs. Although some HP agents, most notably pyruvic acid, can readily freeze into the glass as a neat liquid, a solvent or co-solvent that suppresses ice crystal formation and promotes glassing is required in most cases. Dimethylsulfoxide (DMSO), glycerol and N,N-dimethylacetamide are the most commonly used glassing solvents. Using methods developed for sample vitrification in cryo-electron microscopy, very rapid freezing achieved by spraying droplets into liquid 2-methylbutane at 130K can yield aqueous glasses without needing a vitrifying co-solvent (104), but this technique requires specialized equipment and is not widely used. Choline salts of HP 13C agents and deep eutectic mixtures can have very good glassing, polarization and dissolution characteristics (105). However, depending on the application, the choline may need to be extracted from the solution, and the additional delay may obviate any gain in polarization.
Doping with additional paramagnetic species#
In many cases, ionic paramagnetic dopants can facilitate additional gains in nuclear spin polarization. Small amounts (0.5 - 2 mM) of chelated gadolinium (Gd3+) contrast agents such as gadoteridol (Gd-HP-DO3A) and gadoteric acid (Gd-DOTA) added to the formulation can result in substantially increased polarization, doubled in some cases. The effectiveness varies between different formulations, and it has been attributed to the shortening of the radical electron T1, (106) allowing for more polarization-transferring spin flips with nuclei. Besides Gd3+, this effect has been observed with other paramagnetic lanthanide ions, particularly holmium (Ho3+) (107,108), and also transition metal ions such as manganese (Mn2+). The precise mechanism for the enhancement by paramagnetic ion doping is still a topic of investigation, but recent work with suggests that its effect on the EPR line shape may be more important than the shortening of T1e per se, and the optimal condition is met when the EPR line width matches the 13C nuclear Larmor frequency (109).
Chemical stability#
The highly concentrated solutions and acidic or basic conditions used for some HP agent formulations can affect their chemical stability, and care is required to minimize their degradation. One should consider agent stability when developing a new formulation or when handling ones susceptible to degradation, such as pyruvic acid, glutamine and acetoacetate.
Pyruvic acid and other α-keto acids are light-sensitive and reactive. In liquid state, pyruvic acid will react to form zymonic acid and further oligomers. Parapyruvate also forms readily in pyruvate solutions, and this reaction can even occur in frozen sodium pyruvate solutions stored at -20 ºC (110). If needed, pyruvic acid can be repurified by vacuum distillation, and it should be stored frozen and protected from light. The signals of the degradation products of [1-13C]pyruvic acid have been well characterized (111).
Although most amino acids have excellent chemical stability, glutamine has the tendency to hydrolyze to glutamate and cyclize to pyroglutamate at high or low pH (52). These can potentially interfere with the detection of metabolically-produced [5-13C]glutamate when high-pH formulations are used to formulate glutamine for efficient polarization (53).
Acetoacetate readily decarboxylates to yield acetone and carbon dioxide, particularly under acidic conditions. Acetoacetate must prepared from ester precursors prior to use and handled carefully. The reaction of ethyl [1-13C]acetoacetate with sodium hydroxide to generate sodium [1-13C]acetoacetate yields several impurities, most prominently [1-13C]acetate, which interferes with the reliable detection of the metabolite [5-13C]glutamate
(36,40,112).
Persistent radicals, while relatively stable, are still vulnerable to degradation reactions. They are best stored in a cool, dry environment protected from light. Nitroxyl radicals are quenched under acidic conditions (113), while trityl radicals are quite robust under a range of conditions. To minimize their degradation over extended periods, formulated solutions are best stored frozen at -80 ºC until use. Particularly delicate preparations can be divided up into aliquots for single use, avoiding multiple freeze-thaw cycles.
Formulation development and optimization#
The rationale and general procedure to create an optimized formulation of a HP 13C agent have been well described by Karlsson et al. (114). Formulation development remains an empirical, experimental process, and the following are some general principles and practical suggestions.
Glassing#
When using soluble radicals, it is critical to form a good glass, and higher 13C concentrations facilitate efficient spin diffusion to achieve higher polarization levels faster. The first step then is to identify solvents and conditions that result in a highly-concentrated solution of the target agent which form a glass when rapidly cooled. Fortunately, high solute concentrations often aid glass formation. [Using the unlabeled version of the compound is highly recommended for low-cost screening of glassing conditions.]{.ul} Note that for concentrated solutions, the final volume is substantially affected by the solute, and it will be significantly greater than the added solvent volume. The final volume or density must therefore be measured directly in order to determine the solute concentration.
Glassing can be tested by dipping a vial or tube containing a test solution into liquid nitrogen and seeing whether it remains transparent. A formulation that apparently glasses at the macroscopic level does not necessarily polarize efficiently. A hallmark of poor glass formation is rapid, low solid-state polarization buildup with a non-monoexponential profile (115). Small changes in glassing solvent concentration (~5–10% by volume) can often result in substantially improved polarization performance.
Radical concentration#
The optimal radical concentration will provide the highest polarization in the shortest period of time. Small changes in radical concentration can substantially affect the buildup rate. The buildup dynamics depend on the type of polarizer, and higher radical concentrations are required to achieve the same buildup rates at lower temperature and higher magnetic field strength. Fortunately, higher radical concentrations do not appear to have as great an adverse effect on the maximum polarization as at lower field. If doping with other paramagnetic species, such as Gd3+ chelates, the optimal concentrations are likewise determined empirically.
Microwave frequency#
The microwave frequency for maximum enhancement also needs to be determined for every formulation by performing a microwave sweep (see Hardware for dDNP), and it may vary between preparations, particularly if there is a difference in the radical concentration or other paramagnetic dopant.
Biochemical interactions of HP 13C agents#
Biochemical environment and relaxation dynamics#
As with conventional 1H MRI, the nuclear relaxation rates of 13C are influenced by the environment and can be used to detect changes in oxygenation and distinguish between label resident in different compartments. Although the change in chemical shift is the dominant feature, metabolic transformations of 13C-labeled agents are also accompanied by changes in their relaxation dynamics. Knowledge of metabolite T1s is important for accurately relating their signals to rates of production, but accurately measuring them in the relevant tissues and cellular compartment is not always straight forward, and estimated values obtained in surrogate conditions are often used instead.
The well-known blood-oxygen-level-dependent (BOLD) contrast mechanism, which relies primarily on the T1- and T2*-shortening influence of paramagnetic deoxyhemoglobin in the blood, also affects HP 13C in circulation. A correlation between 1H T2 and 13C-urea T2 has been noted in the rat kidney, and the urea T2 was shorter in kidneys following ischemia-reperfusion (116). The long 13C T2 of [13C,15N2]urea its enables high-resolution imaging, and mapping of T2 relaxation shows anatomically distinct regions in the kidney.
The in vivo 13C T1 is usually estimated on based on in vitro experiments with blood, but experiments in yeast cells show stark differences between extracellular and intracellular T1s. For small carboxylic acids, the intracellular T1s are on the order of <10s (117), and this effect on T1 is also apparent with viscous solutions that mimic the intracellular environment. Serum albumin, the most abundant protein in blood plasma, has also been shown to shorten the 13C T1 of small molecules, particularly those with hydrophobic character (35,118-120).
Transport to sites of metabolic action#
Infused hyperpolarized agents are transported through circulation to the organs and tissues of interest. From there they are taken up by the tissue and in most cases need to cross the cell membrane in order to be metabolized. Delivery and uptake of a hyperpolarized metabolic probe are critical and potentially limiting steps, with delivery in circulation determining the extent of the substrate signal in the tissue of interest, and cellular uptake the maximum possible metabolite signal.
Enzyme kinetics#
By catalyzing specific chemical reactions, enzymes play an essential role in metabolism. The activities of different enzymes are regulated by a variety of mechanisms, but the most basic parameters that determine the rate of an enzyme-catalyzed reaction are: 1) the expression level, or concentration, of the enzyme protein and 2) the substrate concentration. The maximum rate of an enzyme-catalyzed reaction is proportional to the enzyme concentration:
where \(v_{\text{max}}\) is the maximal rate, \(k_{\text{cat}}\) the catalytic efficiency, and [E] the total enzyme concentration. For many enzymes, the relationship between reaction rate (v) and substrate concentration [S] is described by the well-known Michaelis – Menten equation (121):
where \(K_{\text{M}} = \ \frac{k_{\text{off}}\ + \ k_{\text{cat}}}{k_{\text{on}}}\)
and \(k_{\text{off}}\) and \(k_{\text{on}}\) are the rate constants for formation and dissociation of the reactive enzyme-substrate complex (122). In this case, with substrate concentrations much lower than \(K_{\text{M}}\), the rate has an approximately linear dependence. Conversely, the enzyme is saturated with high substrate concentrations and operates at the maximum rate.
Transporter proteins, such as those on the cell surface, also have similar saturable kinetic behavior. They are effectively enzymes, too, that catalyze solute movement across barriers with limited permeability, and flux through a transporter depends on the number of available transporters, their inherent maximum transport rate, and the solute concentration.
Chemical equilibrium#
Many enzyme-catalyzed reactions are readily reversible, particularly when they involve only a small change in free energy, and when the forward and reverse reactions are rapid, a state of equilibrium is established. As an example, the reduction of pyruvate to lactate by lactate dehydrogenase is rapid and reversible.
The relationship between the reactant concentrations at equilibrium is governed by equilibrium constant \(K_{\text{eq}}\):
Biological systems are generally not at thermodynamic equilibrium and an unchanging metabolite concentration usually reflects a steady-state condition instead, where the rates of production and consumption are balanced. However, rapid reversible reactions in a pathway can be near equilibrium when the upstream and downstream reactions are comparatively slow.
Labeled substrates in metabolic pathways#
The use of HP 13C magnetic resonance to probe metabolism is a specialized application of isotope labeling in metabolic research, and it follows the same principles. Despite the tremendous gain in sensitivity that hyperpolarization affords, the short lifetime of the enhanced signal limits the timescale of its use, and it is not a general replacement for other label-detection methods such as mass spectrometry. However, it is the ability of HP 13C to rapidly probe metabolic transformations in intact subjects that distinguishes it from competing methods.
With lower sensitivity than mass spectrometry or radioisotope detection, HP 13C labels are typically used at higher concentrations. This provides a higher substrate fractional enrichment but changes the substrate concentration, and they cannot be treated as tracers. For this reason, most HP experiments can be more accurately considered as the metabolic response to a substrate challenge rather than the direct measurement of the underlying metabolic fluxes. Nonetheless, the data obtained from HP experiments can be analyzed with the established models used for other isotope-labeling methods, with the caveats that the underlying assumptions are often different, and some parameters will rely on estimated values or assumed constraints. With hyperpolarized 13C metabolic data being acquired under pre-steady-state conditions contemporaneous with the delivery of the substrate, along with the variable decay rates of the substrate and metabolite signals makes absolute quantitation of the metabolic fluxes in pre-clinical and clinical settings very challenging. On the other hand, in vitro systems, using isolated enzymes, cell suspensions or flow bioreactors in NMR spectrometers, are more amenable to quantitative metabolic measurements as described in Analysis and Visualization of HP Data.
In either case, when a metabolite is produced by a reversible reaction or is an intermediate in a pathway, the HP 13C metabolite signals will depend strongly not only on the enzymatic fluxes, but also on the endogenous metabolite pool size. Larger pools dilute the labeled metabolites, and the unlabeled metabolites compete in the reactions that consume the metabolite, prolonging the metabolite signal. On the other hand, metabolites produced by irreversible reactions show rates that better reflect the underlying flux. A good example to illustrate these types of reactions is the metabolism of HP [1-13C]pyruvate. Its net conversion to [1-13C]lactate depends not only on lactate dehydrogenase activity but also on its rate of uptake and the intracellular lactate pool size (123), and the substrate concentration can determine whether the transporter or enzyme is the rate-limiting step (124). The conversion of [1-13C]pyruvate to 13CO2 and bicarbonate by pyruvate dehydrogenase is effectively irreversible and the bicarbonate signal corresponds to the enzyme activity (125).
Fractional enrichment and apparent metabolic rate#
The degree to which a HP 13C agent and labeled metabolite(s) are incorporated in the endogenous metabolite pools also plays an important role in determining the apparent metabolic rates. The fractional enrichment describes the amount of labeled metabolite in relation to the sum of the labeled and unlabeled pool. With a small endogenous metabolite pool, a bolus infusion of HP 13C agent can result in a high fractional enrichment and be more quickly metabolized. Pyruvate is normally present in the blood and tissue at much lower concentrations than lactate, and infused HP 13C pyruvate can therefore initially be a significant fraction of the total pyruvate pool. The lactate produced from pyruvate has the same fractional enrichment, but it is diluted by the endogenous lactate, lowering the enrichment. The dilution of the labeled lactate results in its slower conversion back to pyruvate, at least initially. In this way, a large endogenous metabolite pool can act as a sink to slow the consumption of the labeled metabolite and increase its relative signal. The methods used to extract kinetic parameters from HP 13C data are described in Analysis and Visualization of HP Data, with the dynamics of the substrate and metabolite signals used to determine the apparent rates.
Metabolite identification and assignment#
Assigning the spectral peaks produced from the metabolism of an HP 13C agent is an important task, particularly when studying a new agent or target tissue for the first time, and in some cases an unambiguous assignment can be difficult. With limited spectral information – typically the 13C chemical shift alone – additional information and prior knowledge are needed for confident assignments. Even if a spectral signal is well assigned to a particular metabolite, in some cases it may not be clear which particular metabolic pathway or enzyme is responsible for its production.
Metabolite production in other compartments#
One advantage of HP 13C metabolic methods is that the short timescale of the experiment minimizes the contribution of recirculating metabolites to the signals in the organ or tissue of interest. Nonetheless, these effects are not always negligible. The blood makes a small contribution of HP 13C pyruvate-to-lactate conversion (126). Lactate produced from pyruvate elsewhere is part of the signal detected in the rat kidney (127), and splanchnic lactate in the blood is a major contributor to the lactate signal in the heart ventricles (128). Similarly, lactate produced from HP 13C [l]{.smallcaps}- and [d]{.smallcaps}-alanine in the blood are a small fraction of the lactate detected in the kidney (50).
Literature and spectral databases#
Published studies have 13C chemical shift assignments for many metabolites; however, chemical shift values from different sources often do not agree as there are several different referencing conventions in use. Additionally, the conditions used to acquire the data – particularly pH, temperature and concentration – will also influence the chemical shift values obtained. Spectral databases such as the Human Metabolome Database (hmdb.ca) and the Biological Magnetic Resonance Data Bank (bmrb.wisc.edu) are useful resources with extensive collections of metabolite spectra acquired under standardized conditions.
Practical metabolite-assignment experiments#
Several different strategies have been successfully used to assign metabolite signals from HP 13C experiments. In ex vivo experiments, extracts have been prepared from the tissue or cells perfused with 13C-labeled substrate and further analyzed by NMR or liquid chromatography-mass spectrometry (LC-MS) (19). By its sensitivity to interactions between 13C and 1H separated by 2 – 3 chemical bonds, the heteronuclear multibond correlation (HMBC) NMR experiment is particularly well suited for identifying metabolites of HP 13C agents (7,54,129).
Our group has used a number of different methods adapted to the particular substrate and metabolic question to confirm HP 13C metabolite assignments in vivo. Specific labeling can resolve ambiguities arising from a fully-labeled substrates (18). Polarization transfer to neighboring 13C labels can provide additional spectral information (30). Co-polarization and infusion of two different substrates can test whether they yield the same metabolite (35), and specific enzyme inhibitors can be used to test whether a metabolite is produced by a particular pathway (50,56).
Contaminants#
Chemical contaminants from insufficiently-purified preparations or the degradation of sensitive agents are also a source of spurious signals that may be mistaken for metabolites or interfere with the detection and quantitation of real metabolite signals. They are generally identified by their presence in control experiments scanning the HP preparation alone, but differences between in vivo and in vitro conditions such as pH can sometimes complicate their assignment.
Summary and conclusion#
With the physical and metabolic constraints of a lasting HP signal and rapid conversion to detectable products, only a subset of metabolites and metabolic processes are suitable targets for HP 13C agents. Pathways of energy metabolism, particularly glycolysis, the TCA cycle and gluconeogenesis are applicable and sensitive to changes between normal and aberrant metabolism. HP 13C pyruvate remains the most widely used agent, not only for its conversion to lactate in tumors, but also as a metabolic probe in the heart, brain, liver and kidney. A variety of other agents based on carbohydrate, fatty-acid and ketone-body energy metabolites also engage different sections of these pathways, and probes sensitive to pH, tissue necrosis and redox may be particularly applicable for functional imaging of tumors. The ideal characteristics for HP 13C metabolic probes are summarized in Figure 6.8.
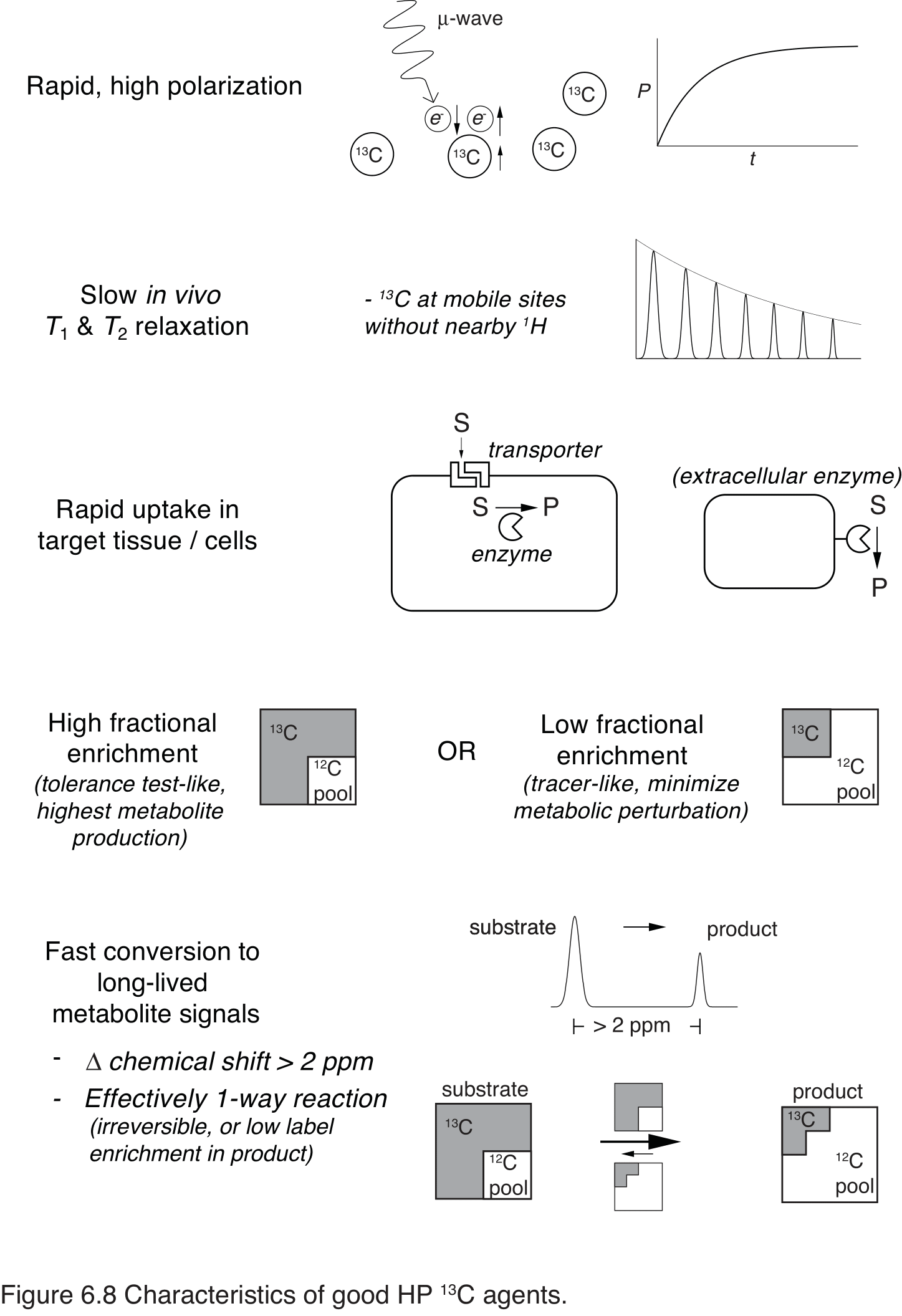
Figure 8 Characteristics of good HP 13C agents. The ideal agent will polarize well and relax slowly in vivo. Uptake in the target compartment will be rapid and specific, with sufficient enrichment and metabolic conversion to detect product. A chemical shift change of >2 ppm is usually required to reliably detect metabolites with the resolutions typically achieved in clinical and pre-clinical scanners. A larger endogenous product pool, such as lactate, dilutes the labeled product, slowing its turnover and prolonging its signal.
With multiple clinical trials with HP 13C pyruvate underway, the method is maturing, and one hopes that they will clearly demonstrate its advantages. In other respects, it remains an emerging technology whose scope of application is still very much being defined. Although the main focus of this chapter is on HP 13C agents with in vivo applications, they and several others have also been used in cell and in vitro applications, where high spectral resolution and control of the experimental conditions enable measurements with higher precision and sensitivity. In vitro and ex vivo methods also serve as the testing ground for new probes. The accrued experience with the metabolic and physicochemical features and constraints particular to HP applications is yielding better designs. Of the myriad possible sites suitable for labeling with HP 13C in the metabolome, most of the evident candidates have been studied, but new metabolic probes sensitive to or selective for particular pathways are still being found, and technical improvements in data acquisition and analysis are enabling more precise and sensitive measurements. With these continuing advances, HP 13C is an increasingly valuable bioanalytical tool and imaging modality.
References#
1. Comment A, Merritt ME. Hyperpolarized magnetic resonance as a sensitive detector of metabolic function. Biochemistry 2014;53:7333–7357. doi: 10.1021/bi501225t.
2. Keshari KR, Wilson DM. Chemistry and biochemistry of (13)C hyperpolarized magnetic resonance using dynamic nuclear polarization. Chem Soc Rev 2014;43:1627–1659. doi: 10.1039/c3cs60124b.
3. Chaumeil MM, Najac C, Ronen SM. Chapter One-Studies of Metabolism Using 13 C MRS of Hyperpolarized Probes. Meth Enzymol 2015;561:1–71. doi: 10.1016/bs.mie.2015.04.001.
4. Brindle KM. Imaging metabolism with hyperpolarized (13)C-labeled cell substrates. J Am Chem Soc 2015;137:6418–6427. doi: 10.1021/jacs.5b03300.
5. Wang ZJ, Ohliger MA, Larson PEZ, Gordon JW, Bok RA, Slater J, Villanueva-Meyer JE, Hess CP, Kurhanewicz J, Vigneron DB. Hyperpolarized 13C MRI: State of the Art and Future Directions. Radiology 2019;291:273–284. doi: 10.1148/radiol.2019182391.
6. Lee P, Leong W, Tan T, Lim M, Han W, Radda GK. In vivo hyperpolarized carbon-13 magnetic resonance spectroscopy reveals increased pyruvate carboxylase flux in an insulin-resistant mouse model. Hepatology 2013;57:515–524. doi: 10.1002/hep.26028.
7. Merritt ME, Harrison C, Sherry AD, Malloy CR, Burgess SC. Flux through hepatic pyruvate carboxylase and phosphoenolpyruvate carboxykinase detected by hyperpolarized 13C magnetic resonance. Proc Natl Acad Sci USA 2011;108:19084–19089. doi: 10.1073/pnas.1111247108.
8. Moreno KX, Moore CL, Burgess SC, Sherry AD, Malloy CR, Merritt ME. Production of hyperpolarized (13)CO2 from [1-(13)C]pyruvate in perfused liver does reflect total anaplerosis but is not a reliable biomarker of glucose production. Metabolomics 2015;11:1144–1156. doi: 10.1007/s11306-014-0768-1.
9. Gallagher FA, Kettunen MI, Hu D-E, et al. Production of hyperpolarized [1,4-13C2]malate from [1,4-13C2]fumarate is a marker of cell necrosis and treatment response in tumors. Proc Natl Acad Sci USA 2009;106:19801–19806. doi: 10.1073/pnas.0911447106.
10. Bohndiek SE, Kettunen MI, Hu DE, Brindle KM. Hyperpolarized 13C Spectroscopy Detects Early Changes in Tumor Vasculature and Metabolism after VEGF Neutralization. Cancer Res 2012;72:854–864. doi: 10.1158/0008-5472.CAN-11-2795.
11. Düwel S, Durst M, Gringeri CV, et al. Multiparametric human hepatocellular carcinoma characterization and therapy response evaluation by hyperpolarized (13) C MRSI. NMR Biomed 2016;29:952–960. doi: 10.1002/nbm.3561.
12. Clatworthy MR, Kettunen MI, Hu D-E, et al. Magnetic resonance imaging with hyperpolarized [1,4-(13)C2]fumarate allows detection of early renal acute tubular necrosis. Proc Natl Acad Sci USA 2012;109:13374–13379. doi: 10.1073/pnas.1205539109.
13. Nielsen PM, Eldirdiri A, Bertelsen LB, Jørgensen HS, Ardenkjaer-Larsen JH, Laustsen C. Fumarase activity: an in vivo and in vitro biomarker for acute kidney injury. Sci Rep 2017;7:17. doi: 10.1038/srep40812.
14. Miller JJ, Lau AZ, Nielsen PM, et al. Hyperpolarized [1,4-13C2]Fumarate Enables Magnetic Resonance-Based Imaging of Myocardial Necrosis. JACC Cardiovasc Imaging 2018;11:1594–1606. doi: 10.1016/j.jcmg.2017.09.020.
15. Keshari KR, Wilson DM, Chen AP, Bok R, Larson PEZ, Hu S, Van Criekinge M, Macdonald JM, Vigneron DB, Kurhanewicz J. Hyperpolarized [2-13C]-fructose: a hemiketal DNP substrate for in vivo metabolic imaging. J Am Chem Soc 2009;131:17591–17596. doi: 10.1021/ja9049355.
16. Allouche-Arnon H, Wade T, Waldner LF, Miller VN, Gomori JM, Katz-Brull R, McKenzie CA. In vivo magnetic resonance imaging of glucose- initial experience. Contrast Media Mol Imaging 2013;8:72–82. doi: 10.1002/cmmi.1497.
17. Rodrigues TB, Serrao EM, Kennedy BWC, Hu D-E, Kettunen MI, Brindle KM. Magnetic resonance imaging of tumor glycolysis using hyperpolarized (13)C-labeled glucose. Nat Med 2013;20:93–97. doi: 10.1038/nm.3416.
18. Mishkovsky M, Anderson B, Karlsson M, Lerche MH, Sherry AD, Gruetter R, Kovacs Z, Comment A. Measuring glucose cerebral metabolism in the healthy mouse using hyperpolarized (13)C magnetic resonance. Sci Rep 2017;7:11719. doi: 10.1038/s41598-017-12086-z.
19. Timm KN, Hartl J, Keller MA, Hu D-E, Kettunen MI, Rodrigues TB, Ralser M, Brindle KM. Hyperpolarized [U-(2) H, U-(13) C]Glucose reports on glycolytic and pentose phosphate pathway activity in EL4 tumors and glycolytic activity in yeast cells. Magn Reson Med 2015;74:1543–1547. doi: 10.1002/mrm.25561.
20. Moreno KX, Harrison CE, Merritt ME, Kovacs Z, Malloy CR, Sherry AD. Hyperpolarized δ-[1-(13) C]gluconolactone as a probe of the pentose phosphate pathway. NMR Biomed 2017;30:e3713. doi: 10.1002/nbm.3713.
21. Moreno KX, Satapati S, Deberardinis RJ, Burgess SC, Malloy CR, Merritt ME. Real-time detection of hepatic gluconeogenic and glycogenolytic states using hyperpolarized [2-13C]dihydroxyacetone. J Biol Chem 2014;289:35859–35867. doi: 10.1074/jbc.M114.613265.
22. Marco-Rius I, Morze von C, Sriram R, et al. Monitoring acute metabolic changes in the liver and kidneys induced by fructose and glucose using hyperpolarized [2-(13) C]dihydroxyacetone. Magn Reson Med 2017;77:65–73. doi: 10.1002/mrm.26525.
23. Marco-Rius I, Cao P, Morze von C, et al. Multiband spectral-spatial RF excitation for hyperpolarized [2-(13) C]dihydroxyacetone (13) C-MR metabolism studies. Magn Reson Med 2016. doi: 10.1002/mrm.26226.
24. Morze von C, Tropp J, Chen AP, et al. Sensitivity enhancement for detection of hyperpolarized 13 C MRI probes with 1 H spin coupling introduced by enzymatic transformation in vivo. Magn Reson Med 2018;80:36–41. doi: 10.1002/mrm.27000.
25. Park JM, Wu M, Datta K, Liu S-C, Castillo A, Lough H, Spielman DM, Billingsley KL. Hyperpolarized Sodium [1-(13)C]-Glycerate as a Probe for Assessing Glycolysis In Vivo. J Am Chem Soc 2017;139:6629–6634. doi: 10.1021/jacs.7b00708.
26. Jensen PR, Peitersen T, Karlsson M, In ‘t Zandt R, Gisselsson A, Hansson G, Meier S, Lerche MH. Tissue-specific short chain fatty acid metabolism and slow metabolic recovery after ischemia from hyperpolarized NMR in vivo. J Biol Chem 2009;284:36077–36082. doi: 10.1074/jbc.M109.066407.
27. Koellisch U, Gringeri CV, Rancan G, Farell EV, Menzel MI, Haase A, Schwaiger M, Schulte RF. Metabolic imaging of hyperpolarized [1-(13) C]acetate and [1-(13) C]acetylcarnitine - investigation of the influence of dobutamine induced stress. Magn Reson Med 2014;74:1011–1018. doi: 10.1002/mrm.25485.
28. Koellisch U, Laustsen C, Nørlinger TS, Ostergaard JA, Flyvbjerg A, Gringeri CV, Menzel MI, Schulte RF, Haase A, Stødkilde-Jørgensen H. Investigation of metabolic changes in STZ-induced diabetic rats with hyperpolarized [1-13C]acetate. Physiol Rep 2015;3. doi: 10.14814/phy2.12474.
29. Bastiaansen JAM, Cheng T, Lei H, Gruetter R, Comment A. Direct noninvasive estimation of myocardial tricarboxylic acid cycle flux in vivo using hyperpolarized C-13 magnetic resonance. J. Mol. Cell. Cardiol. 2015;87:129–137. doi: 10.1016/j.yjmcc.2015.08.012.
30. Mishkovsky M, Comment A, Gruetter R. In vivo detection of brain Krebs cycle intermediate by hyperpolarized magnetic resonance. J Cereb Blood Flow Metab 2012;32:2108–2113. doi: 10.1038/jcbfm.2012.136.
31. Ball DR, Rowlands B, Dodd MS, Le Page L, Ball V, Carr CA, Clarke K, Tyler DJ. Hyperpolarized butyrate: a metabolic probe of short chain fatty acid metabolism in the heart. Magn Reson Med 2014;71:1663–1669. doi: 10.1002/mrm.24849.
32. Bastiaansen JAM, Merritt ME, Comment A. Measuring changes in substrate utilization in the myocardium in response to fasting using hyperpolarized [1-(13)C]butyrate and [1-(13)C]pyruvate. Sci Rep 2016;6:25573. doi: 10.1038/srep25573.
33. Bastiaansen JAM, Yoshihara HAI, Capozzi A, Schwitter J, Gruetter R, Merritt ME, Comment A. Probing cardiac metabolism by hyperpolarized 13C MR using an exclusively endogenous substrate mixture and photo-induced nonpersistent radicals. Magn Reson Med 2018;79:2451–2459. doi: 10.1002/mrm.27122.
34. Abdurrachim D, Teo XQ, Woo CC, Ong SY, Salleh NF, Lalic J, Tan RS, Lee PTH. Cardiac metabolic modulation upon low-carbohydrate low-protein ketogenic diet in diabetic rats studied in vivo using hyperpolarized 13 C pyruvate, butyrate and acetoacetate probes. Diabetes Obes Metab 2019;21:949–960. doi: 10.1111/dom.13608.
35. Yoshihara HAI, Bastiaansen JAM, Karlsson M, Lerche MH, Comment A, Schwitter J. Detection of myocardial medium-chain fatty acid oxidation and tricarboxylic acid cycle activity with hyperpolarized [1-13 C]octanoate. NMR Biomed 2020;33:e4243. doi: 10.1002/nbm.4243.
36. Miller JJ, Ball DR, Lau AZ, Tyler DJ. Hyperpolarized ketone body metabolism in the rat heart. NMR Biomed 2018;60:e3912. doi: 10.1002/nbm.3912.
37. Abdurrachim D, Woo CC, Teo XQ, Chan WX, Radda GK, Lee PTH. A new hyperpolarized 13C ketone body probe reveals an increase in acetoacetate utilization in the diabetic rat heart. Sci Rep 2019;9:5532. doi: 10.1038/s41598-019-39378-w.
38. Abdurrachim D, Teo XQ, Woo CC, Chan WX, Lalic J, Lam CSP, Lee PTH. Empagliflozin reduces myocardial ketone utilization while preserving glucose utilization in diabetic hypertensive heart disease: A hyperpolarized 13 C magnetic resonance spectroscopy study. Diabetes Obes Metab 2018;59:8. doi: 10.1111/dom.13536.
39. Najac C, Radoul M, Le Page LM, Batsios G, Subramani E, Viswanath P, Gillespie AM, Ronen SM. In vivo investigation of hyperpolarized [1,3- 13 C 2 ]acetoacetate as a metabolic probe in normal brain and in glioma. Sci Rep 2019;9:3402. doi: 10.1038/s41598-019-39677-2.
40. Morze von C, Ohliger MA, Marco-Rius I, Wilson DM, Flavell RR, Pearce D, Vigneron DB, Kurhanewicz J, Wang ZJ. Direct assessment of renal mitochondrial redox state using hyperpolarized 13 C-acetoacetate. Magn Reson Med 2018;171:1917. doi: 10.1002/mrm.27054.
41. Hurd RE, Spielman D, Josan S, Yen Y-F, Pfefferbaum A, Mayer D. Exchange-linked dissolution agents in dissolution-DNP (13)C metabolic imaging. Magn Reson Med 2013;70:936–942. doi: 10.1002/mrm.24544.
42. Chen AP, Kurhanewicz J, Bok R, Xu D, Joun D, Zhang V, Nelson SJ, Hurd RE, Vigneron DB. Feasibility of using hyperpolarized [1-13C]lactate as a substrate for in vivo metabolic 13C MRSI studies. Magn Reson Imaging 2008;26:721–726. doi: 10.1016/j.mri.2008.01.002.
43. Morze von C, Chang G-Y, Larson PEZ, et al. Detection of localized changes in the metabolism of hyperpolarized gluconeogenic precursors 13C-lactate and 13C-pyruvate in kidney and liver. Magn Reson Med 2017;77:1429–1437. doi: 10.1002/mrm.26245.
44. Chen AP, Lau JYC, Alvares RDA, Cunningham CH. Using [1-(13) C]lactic acid for hyperpolarized (13) C MR cardiac studies. Magn Reson Med 2015;73:2087–2093. doi: 10.1002/mrm.25354.
45. Bastiaansen JAM, Yoshihara HAI, Takado Y, Gruetter R, Comment A. Hyperpolarized 13C lactate as a substrate for in vivo metabolic studies in skeletal muscle. Metabolomics 2014:1–9.
46. Takado Y, Cheng T, Bastiaansen JAM, Yoshihara HAI, Lanz B, Mishkovsky M, Lengacher S, Comment A. Hyperpolarized 13C Magnetic Resonance Spectroscopy Reveals the Rate-Limiting Role of the Blood-Brain Barrier in the Cerebral Uptake and Metabolism of l-Lactate in Vivo. ACS Chem Neurosci 2018:acschemneuro.8b00066. doi: 10.1021/acschemneuro.8b00066.
47. Ross BD, Hems R, Krebs HA. The rate of gluconeogenesis from various precursors in the perfused rat liver. Biochem J 1967;102:942–951. doi: 10.1042/bj1020942.
48. Exton JH, Park CR. Control of gluconeogenesis in liver. I. General features of gluconeogenesis in the perfused livers of rats. J Biol Chem 1967;242:2622–2636.
49. Park JM, Khemtong C, Liu S-C, Hurd RE, Spielman DM. In vivo assessment of intracellular redox state in rat liver using hyperpolarized [1-13 C]Alanine. Magn Reson Med 2017;77:1741–1748. doi: 10.1002/mrm.26662.
50. Radaelli A, Gruetter R, Yoshihara HAI. In vivo detection of d‐amino acid oxidase with hyperpolarized d‐[1‐13C]alanine. NMR Biomed 2020;33:26941. doi: 10.1002/nbm.4303.
51. Deberardinis RJ, Mancuso A, Daikhin E, Nissim I, Yudkoff M, Wehrli S, Thompson CB. Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci USA 2007;104:19345–19350. doi: 10.1073/pnas.0709747104.
52. Cabella C, Karlsson M, Karlsson M, et al. In vivo and in vitro liver cancer metabolism observed with hyperpolarized [5-13C]glutamine. J Magn Reson 2013;232:45–52. doi: 10.1016/j.jmr.2013.04.010.
53. Xu P, Oosterveer MH, Stein S, et al. LRH-1-dependent programming of mitochondrial glutamine processing drives liver cancer. Genes Dev 2016;30:1255–1260. doi: 10.1101/gad.277483.116.
54. Chaumeil MM, Larson PEZ, Yoshihara HAI, Danforth OM, Vigneron DB, Nelson SJ, Pieper RO, Phillips JJ, Ronen SM. Non-invasive in vivo assessment of IDH1 mutational status in glioma. Nat Commun 2013;4:2429. doi: 10.1038/ncomms3429.
55. Salamanca-Cardona L, Shah H, Poot AJ, et al. In Vivo Imaging of Glutamine Metabolism to the Oncometabolite 2-Hydroxyglutarate in IDH1/2 Mutant Tumors. Cell Metabolism 2017;26:830–841.e3. doi: 10.1016/j.cmet.2017.10.001.
56. Nishihara T, Yoshihara HAI, Nonaka H, et al. Direct Monitoring of γ‐Glutamyl Transpeptidase Activity In Vivo Using a Hyperpolarized 13C‐Labeled Molecular Probe. 2016;55:10626–10629. doi: 10.1002/anie.201603731.
57. Batsios G, Najac C, Cao P, et al. In vivo detection of γ-glutamyl-transferase up-regulation in glioma using hyperpolarized γ-glutamyl-[1-13C]glycine. Sci Rep 2020;10:6244. doi: 10.1038/s41598-020-63160-y.
58. Christensen CE, Karlsson M, Winther JR, Jensen PR, Lerche MH. Non-invasive in-cell determination of free cytosolic [NAD+]/[NADH] ratios using hyperpolarized glucose show large variations in metabolic phenotypes. J Biol Chem 2014;289:2344–2352. doi: 10.1074/jbc.M113.498626.
59. Bohndiek SE, Kettunen MI, Hu D-E, Kennedy BWC, Boren J, Gallagher FA, Brindle KM. Hyperpolarized [1-13C]-ascorbic and dehydroascorbic acid: vitamin C as a probe for imaging redox status in vivo. J Am Chem Soc 2011;133:11795–11801. doi: 10.1021/ja2045925.
60. Keshari KR, Kurhanewicz J, Bok R, Larson PEZ, Vigneron DB, Wilson DM. Hyperpolarized 13C dehydroascorbate as an endogenous redox sensor for in vivo metabolic imaging. Proc Natl Acad Sci USA 2011;108:18606–18611. doi: 10.1073/pnas.1106920108.
61. Timm KN, Hu D-E, Williams M, et al. Assessing Oxidative Stress in Tumors by Measuring the Rate of Hyperpolarized [1-13C]Dehydroascorbic Acid Reduction Using 13C Magnetic Resonance Spectroscopy. J Biol Chem 2017;292:1737–1748. doi: 10.1074/jbc.M116.761536.
62. Qin H, Carroll VN, Sriram R, et al. Imaging glutathione depletion in the rat brain using ascorbate-derived hyperpolarized MR and PET probes. Sci Rep 2018;8:7928. doi: morzemomor.
63. Lippert AR, Keshari KR, Kurhanewicz J, Chang CJ. A hydrogen peroxide-responsive hyperpolarized 13C MRI contrast agent. J Am Chem Soc 2011;133:3776–3779. doi: 10.1021/ja111589a.
64. Wibowo A, Park JM, Liu S-C, Khosla C, Spielman DM. Real-time in vivo detection of H2O2 using hyperpolarized 13C-thiourea. ACS Chem Biol 2017:acschembio.7b00130. doi: 10.1021/acschembio.7b00130.
65. Doura T, Hata R, Nonaka H, Ichikawa K, Sando S. Design of a 13C Magnetic Resonance Probe Using a Deuterated Methoxy Group as a Long-Lived Hyperpolarization Unit. 2012;51:10114–10117. doi: 10.1002/anie.201202885.
66. Nishihara T, Kameyama Y, Nonaka H, Takakusagi Y, Hyodo F, Ichikawa K, Sando S. A Strategy to Design Hyperpolarized (13) C Magnetic Resonance Probes Using [1-(13) C]α-Amino Acid as a Scaffold Structure. Chem Asian J 2017;12:949–953. doi: 10.1002/asia.201700098.
67. Wang S, Korenchan DE, Perez PM, et al. Amino Acid-Derived Sensors for Specific Zn2+ Detection Using Hyperpolarized 13 C Magnetic Resonance Spectroscopy. Chemistry 2019;25:11842–11846. doi: 10.1002/chem.201902771.
68. Mishra A, Pariani G, Oerther T, Schwaiger M, Westmeyer GG. Hyperpolarized Multi-metal (13)C-Sensors for MRI. Anal Chem 2016:acs.analchem.6b03546. doi: 10.1021/acs.analchem.6b03546.
69. Gallagher FA, Kettunen MI, Day SE, et al. Magnetic resonance imaging of pH in vivo using hyperpolarized 13C-labelled bicarbonate. Nature 2008;453:940–943. doi: 10.1038/nature07017.
70. Scholz DJ, Otto AM, Hintermair J, et al. Parameterization of hyperpolarized (13)C-bicarbonate-dissolution dynamic nuclear polarization. Magn Reson Mater Phy 2015;28:591–598. doi: 10.1007/s10334-015-0500-9.
71. Schroeder MA, Swietach P, Atherton HJ, Gallagher FA, Lee P, Radda GK, Clarke K, Tyler DJ. Measuring intracellular pH in the heart using hyperpolarized carbon dioxide and bicarbonate: a 13C and 31P magnetic resonance spectroscopy study. Cardiovasc Res 2010;86:82–91. doi: 10.1093/cvr/cvp396.
72. Lau AZ, Miller JJ, Tyler DJ. Mapping of intracellular pH in the in vivo rodent heart using hyperpolarized [1-13C]pyruvate. Magn Reson Med 2017;77:1810–1817. doi: 10.1002/mrm.26260.
73. Ghosh RK, Kadlecek SJ, Pourfathi M, Rizi RR. Efficient production of hyperpolarized bicarbonate by chemical reaction on a DNP precursor to measure pH. Magn Reson Med 2014;74:1406–1413. doi: 10.1002/mrm.25530.
74. Korenchan DE, Flavell RR, Baligand C, Sriram R, Neumann K, Sukumar S, VanBrocklin H, Vigneron DB, Wilson DM, Kurhanewicz J. Dynamic nuclear polarization of biocompatible 13C-enriched carbonates for in vivo pH imaging. Chem. Commun. 2016;52:3030–3033. doi: 10.1039/C5CC09724J.
75. Hundshammer C, Düwel S, Ruseckas D, et al. Hyperpolarized Amino Acid Derivatives as Multivalent Magnetic Resonance pH Sensor Molecules. Sensors-Basel 2018;18:600. doi: 10.3390/s18020600.
76. Flavell RR, Morze von C, Blecha JE, et al. Application of Good’s buffers to pH imaging using hyperpolarized (13)C MRI. Chem Commun (Camb) 2015;51:14119–14122. doi: 10.1039/c5cc05348j.
77. Korenchan DE, Taglang C, Morze von C, et al. Dicarboxylic acids as pH sensors for hyperpolarized (13)C magnetic resonance spectroscopic imaging. Analyst 2017;142:1429–1433. doi: 10.1039/c7an00076f.
78. Düwel S, Hundshammer C, Gersch M, et al. Imaging of pH in vivo using hyperpolarized (13)C-labelled zymonic acid. Nat Commun 2017;8:15126. doi: 10.1038/ncomms15126.
79. Svensson J, Månsson S, Johansson E, Petersson JS, Olsson LE. Hyperpolarized 13C MR angiography using trueFISP. Magn Reson Med 2003;50:256–262. doi: 10.1002/mrm.10530.
80. Kettunen MI, Kennedy BWC, Hu D-E, Brindle KM. Spin echo measurements of the extravasation and tumor cell uptake of hyperpolarized [1-(13) C]lactate and [1-(13) C]pyruvate. Magn Reson Med 2013;70:1200–1209. doi: 10.1002/mrm.24591.
81. Morze von C, Larson PEZ, Hu S, Yoshihara HAI, Bok RA, Goga A, Ardenkjaer-Larsen JH, Vigneron DB. Investigating tumor perfusion and metabolism using multiple hyperpolarized (13)C compounds: HP001, pyruvate and urea. Magn Reson Imaging 2012;30:305–311. doi: 10.1016/j.mri.2011.09.026.
82. Grant AK, Vinogradov E, Wang X, Lenkinski RE, Alsop DC. Perfusion imaging with a freely diffusible hyperpolarized contrast agent. Magn Reson Med 2011;66:746–755. doi: 10.1002/mrm.22860.
83. Cheng T, Mishkovsky M, Junk MJN, Münnemann K, Comment A. Producing Radical-Free Hyperpolarized Perfusion Agents for In Vivo Magnetic Resonance Using Spin-Labeled Thermoresponsive Hydrogel. Macromol Rapid Commun 2016;37:1074–1078. doi: 10.1002/marc.201600133.
84. Morze von C, Bok RA, Reed GD, Ardenkjaer-Larsen JH, Kurhanewicz J, Vigneron DB. Simultaneous multiagent hyperpolarized (13)C perfusion imaging. Magn Reson Med 2014;72:1599–1609. doi: 10.1002/mrm.25071.
85. Ardenkjaer-Larsen JH, Fridlund B, Gram A, Hansson G, Hansson L, Lerche MH, Servin R, Thaning M, Golman K. Increase in signal-to-noise ratio of > 10,000 times in liquid-state NMR. P Natl Acad Sci Usa 2003;100:10158–10163. doi: 10.1073/pnas.1733835100.
86. Golman K, Ardenkjaer-Larsen JH, Petersson JS, Månsson S, Leunbach I. Molecular imaging with endogenous substances. P Natl Acad Sci Usa 2003;100:10435–10439. doi: 10.1073/pnas.1733836100.
87. Morze von C, Bok RA, Sands JM, Kurhanewicz J, Vigneron DB. Monitoring urea transport in rat kidney in vivo using hyperpolarized 13C magnetic resonance imaging. Am J Physiol Renal Physiol 2012;302:F1658–F1662. doi: 10.1152/ajprenal.00640.2011.
88. Bertelsen LB, Nielsen PM, Qi H, Nørlinger TS, Zhang X, Stødkilde-Jørgensen H, Laustsen C. Diabetes induced renal urea transport alterations assessed with 3D hyperpolarized (13) C,(15) N-Urea. Magn Reson Med 2016;77:1650–1655. doi: 10.1002/mrm.26256.
89. Reed GD, Morze von C, Bok R, Koelsch BL, Van Criekinge M, Smith KJ, Hong Shang, Larson PEZ, Kurhanewicz J, Vigneron DB. High resolution (13)C MRI with hyperpolarized urea: in vivo T(2) mapping and (15)N labeling effects. IEEE Trans Med Imaging 2014;33:362–371. doi: 10.1109/TMI.2013.2285120.
90. Reed GD, Morze von C, Verkman AS, et al. Imaging Renal Urea Handling in Rats at Millimeter Resolution using Hyperpolarized Magnetic Resonance Relaxometry. Tomography 2016;2:125–135. doi: 10.18383/j.tom.2016.00127.
91. Wolber J, Ellner F, Fridlund B, Gram A, Johannesson H, Hansson G, Hansson LH, Lerche MH, Månsson S, Servin R. Generating highly polarized nuclear spins in solution using dynamic nuclear polarization. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment 2004;526:173–181. doi: 10.1016/j.nima.2004.03.171.
92. Lumata L, Ratnakar SJ, Jindal A, Merritt M, Comment A, Malloy C, Sherry AD, Kovacs Z. BDPA: an efficient polarizing agent for fast dissolution dynamic nuclear polarization NMR spectroscopy. Chemistry 2011;17:10825–10827. doi: 10.1002/chem.201102037.
93. Mccarney ER, Armstrong BD, Kausik R, Han S. Dynamic nuclear polarization enhanced nuclear magnetic resonance and electron spin resonance studies of hydration and local water dynamics in micelle and vesicle assemblies. Langmuir : the ACS journal of surfaces and colloids 2008;24:10062–10072. doi: 10.1021/la800334k.
94. Lingwood MD, Siaw TA, Sailasuta N, Ross BD, Bhattacharya P, Han S. Continuous flow Overhauser dynamic nuclear polarization of water in the fringe field of a clinical magnetic resonance imaging system for authentic image contrast. J Magn Reson 2010;205:247–254. doi: 10.1016/j.jmr.2010.05.008.
95. Gajan D, Bornet A, Vuichoud B, et al. Hybrid polarizing solids for pure hyperpolarized liquids through dissolution dynamic nuclear polarization. Proc Natl Acad Sci USA 2014;111:14693–14697. doi: 10.1073/pnas.1407730111.
96. Miéville P, Ahuja P, Sarkar R, et al. Scavenging free radicals to preserve enhancement and extend relaxation times in NMR using dynamic nuclear polarization. 2010;49:6182–6185. doi: 10.1002/anie.201000934.
97. Cheng T, Mishkovsky M, Bastiaansen JAM, Ouari O, Hautle P, Tordo P, van den Brandt B, Comment A. Automated transfer and injection of hyperpolarized molecules with polarization measurement prior to in vivo NMR. NMR Biomed 2013;26:1582–1588. doi: 10.1002/nbm.2993.
98. Guzmán MI, Colussi AJ, Hoffmann MR. Photogeneration of distant radical pairs in aqueous pyruvic acid glasses. J Phys Chem A 2006;110:931–935. doi: 10.1021/jp053449t.
99. Eichhorn TR, Takado Y, Salameh N, Capozzi A, Cheng T, Hyacinthe J-N, Mishkovsky M, Roussel C, Comment A. Hyperpolarization without persistent radicals for in vivo real-time metabolic imaging. Proc Natl Acad Sci USA 2013;110:18064–18069. doi: 10.1073/pnas.1314928110.
100. Capozzi A, Karlsson M, Petersen JR, Lerche MH, Ardenkjaer-Larsen JH. Liquid-State 13C Polarization of 30% through Photoinduced Nonpersistent Radicals. J. Phys. Chem. C 2018:acs.jpcc.8b01482. doi: 10.1021/acs.jpcc.8b01482.
101. Capozzi A, Cheng T, Boero G, Roussel C, Comment A. Thermal annihilation of photo-induced radicals following dynamic nuclear polarization to produce transportable frozen hyperpolarized (13)C-substrates. Nat Commun 2017;8:15757. doi: 10.1038/ncomms15757.
102. Marco-Rius I, Cheng T, Gaunt AP, Patel S, Kreis F, Capozzi A, Wright AJ, Brindle KM, Ouari O, Comment A. Photogenerated Radical in Phenylglyoxylic Acid for in Vivo Hyperpolarized 13C MR with Photosensitive Metabolic Substrates. J Am Chem Soc 2018;140:14455–14463. doi: 10.1021/jacs.8b09326.
103. Capozzi A, Patel S, Gunnarsson CP, Marco-Rius I, Comment A, Karlsson M, Lerche MH, Ouari O, Ardenkjaer-Larsen JH. Efficient Hyperpolarization of U-13 C-Glucose Using Narrow-Line UV-Generated Labile Free Radicals. 2019;58:1334–1339. doi: 10.1002/anie.201810522.
104. Lama B, Collins JHP, Downes D, Smith AN, Long JR. Expeditious dissolution dynamic nuclear polarization without glassing agents. NMR Biomed 2016;29:226–231. doi: 10.1002/nbm.3473.
105. Bowen S, Ardenkjaer-Larsen JH. Formulation and utilization of choline based samples for dissolution dynamic nuclear polarization. J Magn Reson 2013;236:26–30. doi: 10.1016/j.jmr.2013.08.007.
106. Jóhannesson H, Macholl S, Ardenkjaer-Larsen JH. Dynamic Nuclear Polarization of [1-13C]pyruvic acid at 4.6 tesla. J Magn Reson 2009;197:167–175. doi: 10.1016/j.jmr.2008.12.016.
107. Gordon JW, Fain SB, Rowland IJ. Effect of lanthanide ions on dynamic nuclear polarization enhancement and liquid-state T1 relaxation. Magn Reson Med 2012;68:1949–1954. doi: 10.1002/mrm.24207.
108. Khattri RB, Sirusi AA, Suh EH, Kovacs Z, Merritt ME. The influence of Ho3+ doping on 13C DNP in the presence of BDPA. Phys Chem Chem Phys 2019;21:18629–18635. doi: 10.1039/c9cp03717a.
109. Capozzi A, Patel S, Wenckebach WT, Karlsson M, Lerche MH, Ardenkjaer-Larsen JH. Gadolinium Effect at High-Magnetic-Field DNP: 70% 13C Polarization of [U- 13C] Glucose Using Trityl. J. Phys. Chem. Lett. 2019;10:3420–3425. doi: 10.1021/acs.jpclett.9b01306.
110. Margolis SA, Coxon B. Identification and quantitation of the impurities in sodium pyruvate. Anal Chem 1986;58:2504–2510. doi: 10.1021/ac00125a033.
111. Harris T, Gamliel A, Sosna J, Gomori JM, Katz-Brull R. Impurities of [1-13C]Pyruvic Acid and a Method to Minimize Their Signals for Hyperpolarized Pyruvate Metabolism Studies. Appl Magn Reson 2018;49:1085–1098. doi: 10.1007/s00723-018-1030-1.
112. Chen W, Sharma G, Jiang W, Maptue NR, Malloy CR, Sherry AD, Khemtong C. Metabolism of hyperpolarized 13 C-acetoacetate to β-hydroxybutyrate detects real-time mitochondrial redox state and dysfunction in heart tissue. NMR Biomed 2019;32:e4091. doi: 10.1002/nbm.4091.
113. Sen VD, Golubev VA. Kinetics and mechanism for acid‐catalyzed disproportionation of 2,2,6,6‐tetramethylpiperidine‐1‐oxyl. Journal of Physical Organic Chemistry 2009;22:138–143. doi: 10.1002/poc.1439.
114. Karlsson M, Jensen PR, Duus JØ, Meier S, Lerche MH. Development of dissolution DNP-MR substrates for metabolic research. Appl Magn Reson 2012;43:223–236. doi: 10.1007/s00723-012-0336-7.
115. Flori A, Giovannetti G, Santarelli MF, Aquaro GD, De Marchi D, Burchielli S, Frijia F, Positano V, Landini L, Menichetti L. Biomolecular imaging of 13C-butyrate with dissolution-DNP: Polarization enhancement and formulation for in vivo studies. Spectrochim Acta A 2018;199:153–160. doi: 10.1016/j.saa.2018.03.014.
116. Mariager CØ, Nielsen PM, Qi H, Ringgaard S, Laustsen C. Hyperpolarized 13C,15N2‐urea T2 relaxation changes in acute kidney injury. Magn Reson Med 2018;80:696–702. doi: 10.1002/mrm.27050.
117. Karlsson M, Jensen PR, Ardenkjaer-Larsen JH, Lerche MH. Difference between Extra- and Intracellular T1 Values of Carboxylic Acids Affects the Quantitative Analysis of Cellular Kinetics by Hyperpolarized NMR. 2016;55:13567–13570. doi: 10.1002/anie.201607535.
118. Moreno KX, Sabelhaus SM, Merritt ME, Sherry AD, Malloy CR. Competition of pyruvate with physiological substrates for oxidation by the heart: implications for studies with hyperpolarized [1-13C]pyruvate. Am J Physiol Heart Circ Physiol 2010;298:H1556–64. doi: 10.1152/ajpheart.00656.2009.
119. Lerche MH, Meier S, Jensen PR, Baumann H, Petersen BO, Karlsson M, Duus JØ, Ardenkjaer-Larsen JH. Study of molecular interactions with 13C DNP-NMR. J Magn Reson 2010;203:52–56. doi: 10.1016/j.jmr.2009.11.020.
120. Keshari KR, Kurhanewicz J, Macdonald JM, Wilson DM. Generating contrast in hyperpolarized 13C MRI using ligand-receptor interactions. Analyst 2012;137:3427–3429. doi: 10.1039/c2an35406c.
121. Menten L, Michaelis MI. Die kinetik der invertinwirkung. Biochem Z 1913;49:5.
122. Briggs GE, Haldane JBS. A Note on the Kinetics of Enzyme Action. Biochem J 1925;19:338–339. doi: 10.1042/bj0190338.
123. Day SE, Kettunen MI, Gallagher FA, Hu D-E, Lerche M, Wolber J, Golman K, Ardenkjaer-Larsen JH, Brindle KM. Detecting tumor response to treatment using hyperpolarized 13C magnetic resonance imaging and spectroscopy. Nat Med 2007;13:1382–1387. doi: 10.1038/nm1650.
124. Witney TH, Kettunen MI, Brindle KM. Kinetic modeling of hyperpolarized 13C label exchange between pyruvate and lactate in tumor cells. J Biol Chem 2011;286:24572–24580. doi: 10.1074/jbc.M111.237727.
125. Schroeder MA, Atherton HJ, Heather LC, Griffin JL, Clarke K, Radda GK, Tyler DJ. Determining the in vivo regulation of cardiac pyruvate dehydrogenase based on label flux from hyperpolarised [1-13C]pyruvate. NMR Biomed 2011;24:980–987. doi: 10.1002/nbm.1668.
126. Allouche-Arnon H, Lerche MH, Karlsson M, Lenkinski RE, Katz-Brull R. Deuteration of a molecular probe for DNP hyperpolarization–a new approach and validation for choline chloride. Contrast Media Mol Imaging 2011;6:499–506. doi: 10.1002/cmmi.452.
127. Chen AP, Leung K, Lam W, Hurd RE, Vigneron DB, Cunningham CH. Design of spectral-spatial outer volume suppression RF pulses for tissue specific metabolic characterization with hyperpolarized 13C pyruvate. J Magn Reson 2009;200:344–348. doi: 10.1016/j.jmr.2009.06.021.
128. Wespi P, Steinhauser J, Kwiatkowski G, Kozerke S. Overestimation of cardiac lactate production caused by liver metabolism of hyperpolarized [1-13 C]pyruvate. Magn Reson Med 2018;100:10158. doi: 10.1002/mrm.27197.
129. Schroeder MA, Atherton HJ, Ball DR, Cole MA, Heather LC, Griffin JL, Clarke K, Radda GK, Tyler DJ. Real-time assessment of Krebs cycle metabolism using hyperpolarized 13C magnetic resonance spectroscopy. FASEB J. 2009;23:2529–2538. doi: 10.1096/fj.09-129171.