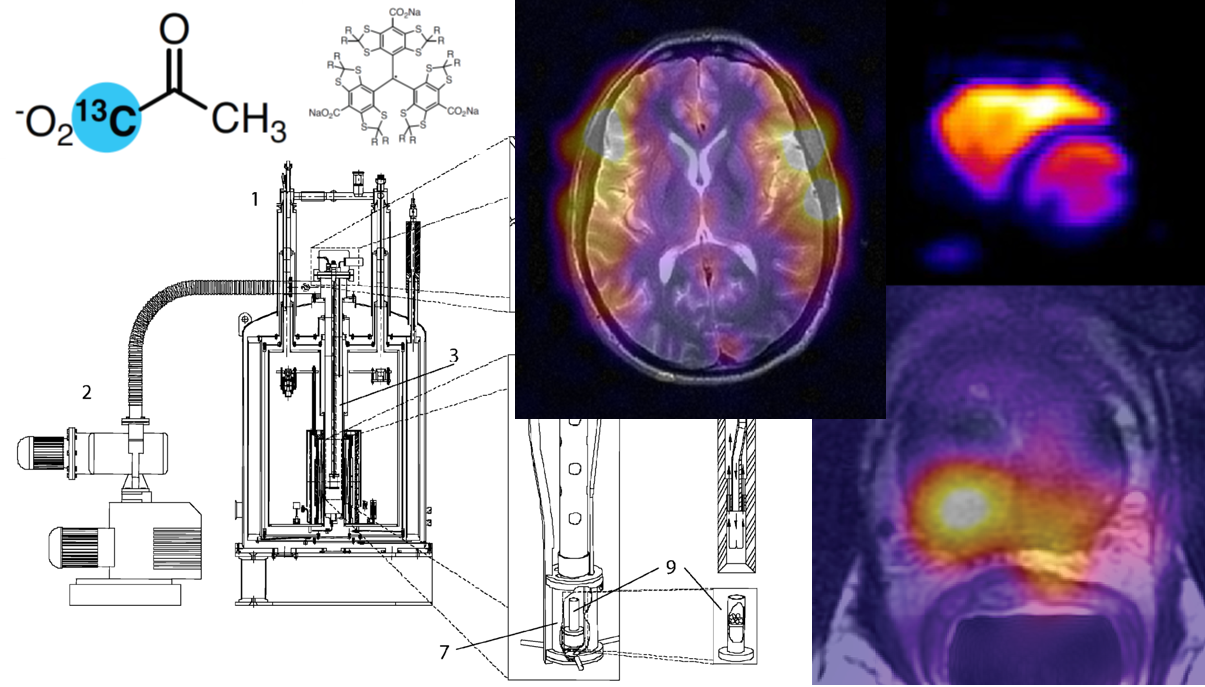
Integration into Liver Studies#
Cornelius von Morze, PhD, Assistant Professor, Mallinckrodt Institute of Radiology, Washington University
Michael A. Ohliger, MD PhD, Associate Professor, Department of Radiology and Biomedical Imaging, University of California, San Francisco
Abstract
Hyperpolarized (HP) 13C MRI can provide non-invasive metabolic imaging readouts along several key hepatic metabolic pathways, including pyruvate-lactate exchange, glutaminolysis, gluconeogenesis, and the metabolism of ketones and short chain fatty acids. Integration of HP 13C MRI methods into liver imaging studies introduces various specialized technical challenges, due to its anatomic location within the abdomen and associated respiratory motion. These challenges can be addressed by rapid imaging strategies such as metabolite-specific spectral-spatial (SPSP) radiofrequency excitation with echo planar imaging (EPI) readout, which is also well suited for the rapid T1 decay of HP magnetization. Hyperpolarized 13C MRI based on [1-13C]pyruvate and a variety of other probes is applicable for the study of pathophysiologic metabolic changes in primary liver cancer, mainly hepatocellular carcinoma (HCC), as well as metastatic disease. Proof of principle clinical translation has recently been demonstrated for imaging metastatic liver lesions using [1-13C]pyruvate. [1-13C]pyruvate and other probes are also applicable to the study of diffuse liver disease, especially non-alcoholic steatohepatitis (NASH).
Keywords: Liver, Hyperpolarized, 13C, MRI, HCC, NASH
Liver imaging: state of the art#
The primary modalities for medical imaging of the liver are ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI). These scans are routinely used to assess liver size and contour, to detect and evaluate focal lesions, and to evaluate vascular and biliary anatomy. Although these applications are extremely important and useful, they reflect a traditional radiological view of the liver as a static lump of tissue (1), belying its highly dynamic physiologic functions, especially the rich metabolic processes occurring below the surface. More recently, sophisticated variations on these modalities have been developed to provide added deeper insights into liver disease, especially diffuse liver disease. For example, proton density fat fraction (MRI-PDFF), a form of localized 1H MR spectroscopy, has emerged as the gold standard approach for quantifying hepatic steatosis (2). Supplemented by mechanical shear wave excitation, US- and MRI- elastography can be used to measure liver stiffness as a marker for liver fibrosis (3). Molecular imaging has thus far made only a relatively minor impact on the clinical management of patients with primary liver disease. A notable exception is gadoxetate MRI, which can be used to probe the activity of OATP transporters in hepatocytes, and is routinely applied for evaluating primary as well as metastatic liver lesions (4). Although frequently used to detect liver metastases (5), the role of positron emission tomography (PET), the gold standard of molecular imaging, has been somewhat limited in the context of primary liver disease. This might be because the liver’s rich metabolic functions tend to accumulate labels fairly non-specifically, with normal liver typically appearing bright with many different small molecule tracers.
Hyperpolarized (HP) 13C MRI has emerged as a viable, clinically translatable, approach for imaging the liver, with the potential to probe important hepatic physiologic processes non-invasively including perfusion, transport, and especially metabolism, by exploiting the unparalleled specificity of 13C MR for metabolic pathways of interest. In particular, hyperpolarized [1-13C]pyruvate is rapidly metabolized into [1-13C]lactate and [1-13C]alanine in liver tissue, via lactate dehydrogenase (LDH) and alanine transaminase (ALT), respectively, providing a new set of potential biomarkers of liver disease. As reviewed in detail below, HP [1-13C]pyruvate and numerous other HP 13C probes have been applied for studies of liver pathophysiology.
Compared to PET imaging, HP 13C MRI has several potential advantages in liver imaging. In HP 13C, several probes can be imaged at once, offering simultaneous assessment of multiple metabolic pathways. In addition, PET relies on the accumulation of a radiotracer at a target site. Uptake of 18F-fluorodeoxyglucose (FDG) in hepatocellular carcinoma (HCC), for example, is influenced by the expression of glucose-6-phosphatase (6), which permits the release of trapped FDG. Because HP 13C is sensitive to metabolic changes it requires no such trapping.
Technical challenges of liver HP 13C MRI#
Besides the demanding logistical requirements universally associated with the short signal lifetime of HP magnetization, HP liver imaging presents a unique set of additional technical challenges. As depicted in Figure 1, the dual vascular input to the liver from the portal vein (major source of perfusion) and hepatic artery could complicate the interpretation of signal dynamics. In addition, because HP material in the portal vein has already passed through the GI tract, it could potentially have a different profile than that originating from the hepatic artery. Finally, it is well-known that dramatic alterations in the relative blood flow in the hepatic artery and portal vein can occur with meals, and these changes are further modulated by the presence of superimposed liver diseases such as portal hypertension (7).
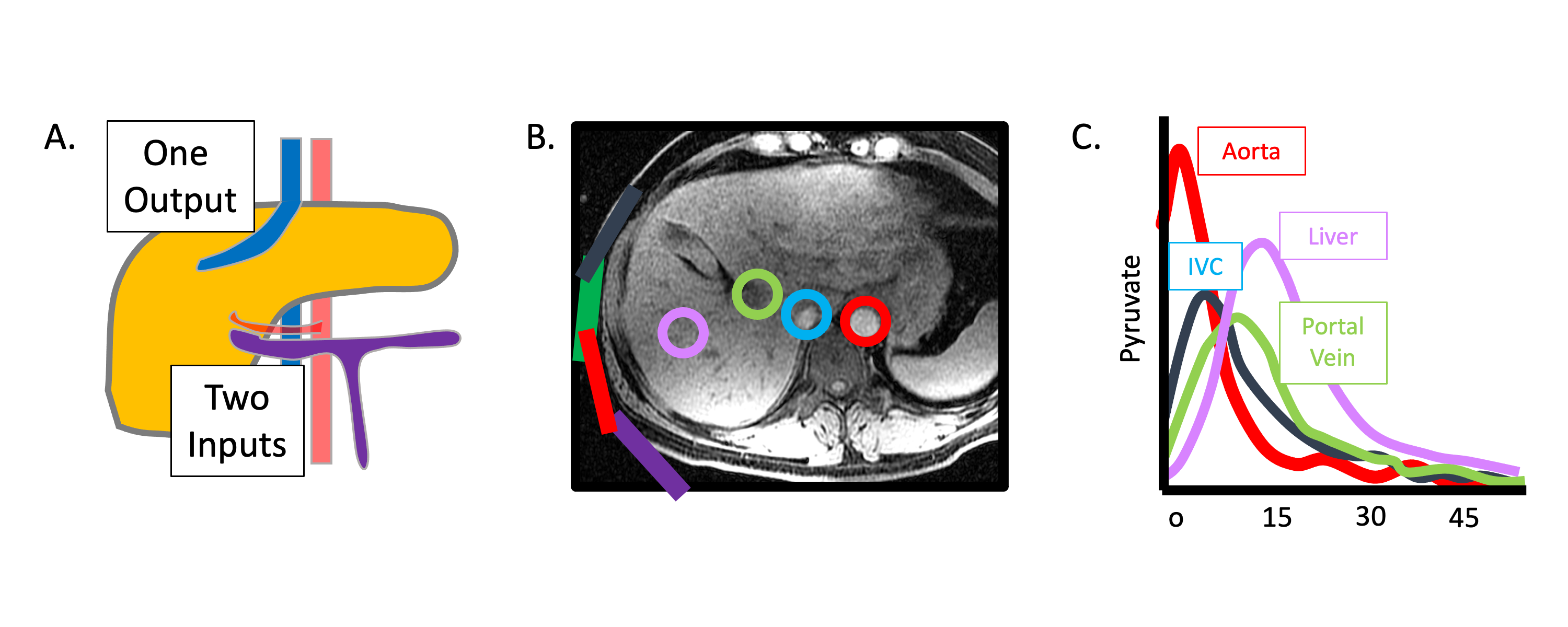
Figure 1: Dual blood supply of the liver visualized using HP 13C MRI. (A) Schematic illustration of the blood supply of the liver, showing the portal vein (purple) draining the gastrointestinal tract and the hepatic artery (red), with a single output hepatic vein (blue). (B) T1-weighted image of the liver with ROIs drawn around the aorta (red), inferior vena cava (blue), portal vein (green), and liver parenchyma (purple). (C) HP [1-13C]pyruvate signal measured over time from the vessels shown in (B) demonstrates clear distinct inputs from the arterial and portal venous supply
Another challenge in imaging the liver with HP 13C is breathing motion. Most dynamic HP acquisitions (40-60 s) are too long for a single breath-hold. The large respiratory motion of the liver demands especially fast image acquisition, ideally triggered by respiratory signals. Echo planar imaging (EPI) with metabolite-specific spectral-spatial (SPSP) radiofrequency excitation is an appealing fast imaging strategy for HP 13C MRI of the liver (8-10), but can be vulnerable to B0 inhomogeneity (Figure 2), with typical SPSP pass- and stop-band widths as small as +/- 0.5 ppm. Errors in the SPSP pulse center frequency can lead to application of incorrect flip angles and/or image reconstruction artifacts, potentially confounding data interpretation. Some degree of frequency offset is likely inevitable due to higher B0 inhomogeneity in the abdomen as compared with more stationary and uniform tissues like brain.

Figure 2 Field map (Hz) in the abdomen acquired using the IDEAL-IQ multiecho gradient echo method at 3T shows large B0 frequency variation in this slice, which can be a challenge for frequency-selective acquisition techniques. Colorbar represents frequency offset in hertz at 3T.
Intrinsic tissue properties of the liver may also present challenges. The presence of bowel gas within the gastrointestinal tract can create large magnetic field variations within the abdomen caused by magnetic susceptibility. In addition, 13C T1 and T2 relaxation times may also be shorter in liver than other issues, due to paramagnetic relaxation by liver iron and/or other factors (11).
In terms of acquisition hardware phased array reception can provide increased SNR (at least peripherally) and the coverage that is needed to cover the abdomen (12), with the added potential for parallel imaging (13-15). Such approaches have helped revolutionize body 1H MRI, but new methods are needed for parallel 13C reconstructions based on limited availability of calibration data (16,17). The ideal size and number of coils for 13C imaging of the abdomen is not yet clear, but should be similar to 1H imaging at the same Larmor frequency. At the same field strength, therefore, the optimal number of coil elements for a 13C -sensitive coil array is likely less than that for a proton array.
A large transmitter coil is necessary to provide high transmit RF homogeneity over the abdomen, with a clamshell insert transmitter coil having been used for initial studies (18). However, such a coil occupies a significant fraction of the scanner bore, placing a significant constraint on patient size. In this regard, abdominal imaging would stand to benefit tremendously from the development of a dual-tuned 13C /1H body coil to handle 13C excitation (19). Likewise, a significant appeal of 1H-based detection of HP 13C probes following polarization transfer (20,21) is the potential to liberate space inside the scanner bore occupied by 13C receiver coils.
Liver metabolic pathways accessible via HP imaging#
Numerous studies have investigated liver metabolism using HP [1-13C]pyruvate, following initial work showing the potential to probe oxidative and anaplerotic pathways of pyruvate metabolism in perfused liver (22), as well as exchange into [1-13C]lactate and [1-13C]alanine, which constitute the major hepatic signals observed in vivo. Not surprisingly, exchange into these dynamic hepatic metabolite pools has been shown to be influenced by nutritional status (fasted vs normal fed) (23), as well as by insulin deficiency and insulin resistance (24,25). Hepatic HP [1-13C]lactate metabolite signals are also sensitive to drug treatments such as metformin (26), which arrests hepatic mitochondrial metabolism at the level of complex I, altering the cytosolic NAD(H) redox state. Alternatively HP [2-13C]pyruvate can be used to probe TCA intermediates, although normal hepatic flux through pyruvate dehydrogenase (PDH) is low unless stimulated acutely such as by dichloroacetate treatment (27).
Several hepatic metabolic pathways accessible via HP 13C MRI are illustrated in Figure 3. Unlike many tumor tissues and brain, which are common targets for HP 13C imaging and rely largely on glucose to meet their energetic demands, normal liver relies predominantly on fatty acids for energy to support its myriad biosynthetic functions, suggesting that metabolic probes other than pyruvate might be useful. As illustrated in Figure 3, other potentially relevant probes of liver metabolism include the short acid fatty acid [1-13C]butyrate (28) and ketone body [1,3-13C2]acetoacetate (29), both of which could be used to assess hepatic mitochondrial metabolism. Fatty acids with chain lengths longer than four carbon units generally have insufficient T1’s and/or cellular uptake for HP 13C metabolic imaging studies. HP [2-13C]dihydroxyacetone (30) and [2-13C]fructose (31) are rapidly taken up and phosphorylated by liver, offering potential assessment of liver energy charge that can complement the redox-based assessment offered by pyruvate. These substrates introduce bidirectional 13C labeling upstream of pyruvate along the pathways of glycolysis / gluconeogenesis. Direct detection of glucose production is possible using 1H-decoupled HP [2-13C]dihydroxyacetone (30,32).
Conversion of [1-13C]dehydroascorbate to [1-13C]vitamin C (33) can be used to directly probe hepatic oxidative stress, providing a complementary view of redox as mediated by NADP(H) as compared to NAD(H). [1-13C]alanine has been used to directly probe the hepatic intracellular lactate / pyruvate ratio (34,35), as the alanine rapidly exchanges with lactate and pyruvate once it enters the cell, providing a means to discriminate intracellular signals from larger extracellular signals. The lower cellular permeability of alanine as compared to pyruvate has been addressed by use of an ethyl ester (36). Lactate, as the dominant circulating substrate as compared to pyruvate, has also been used for HP 13C studies, but label exchange into pyruvate is much lower than when injecting pyruvate, because of the smaller pyruvate pool size. Flux through the pentose phosphate pathway (PPP) has been assessed in perfused livers using HP \(\delta\)-[1-13C]gluconolactone. Additionally, HP [13C]urea and other metabolically inert probes can be used to assess liver perfusion and vascular permeability (37,38).
15N probes such as L-[15N]carnitine can provide longer signal lifetimes for HP liver imaging, which remains feasible for at least three minutes after 15N injection (39). Finally, the liver-specific paramagnetic relaxation agent gadoxetate together with hyperpolarization offers an avenue toward hepatocyte-specific metabolic measurements, by quenching extraneous HP signals including those arising from adjacent vasculature or other cell types (40).
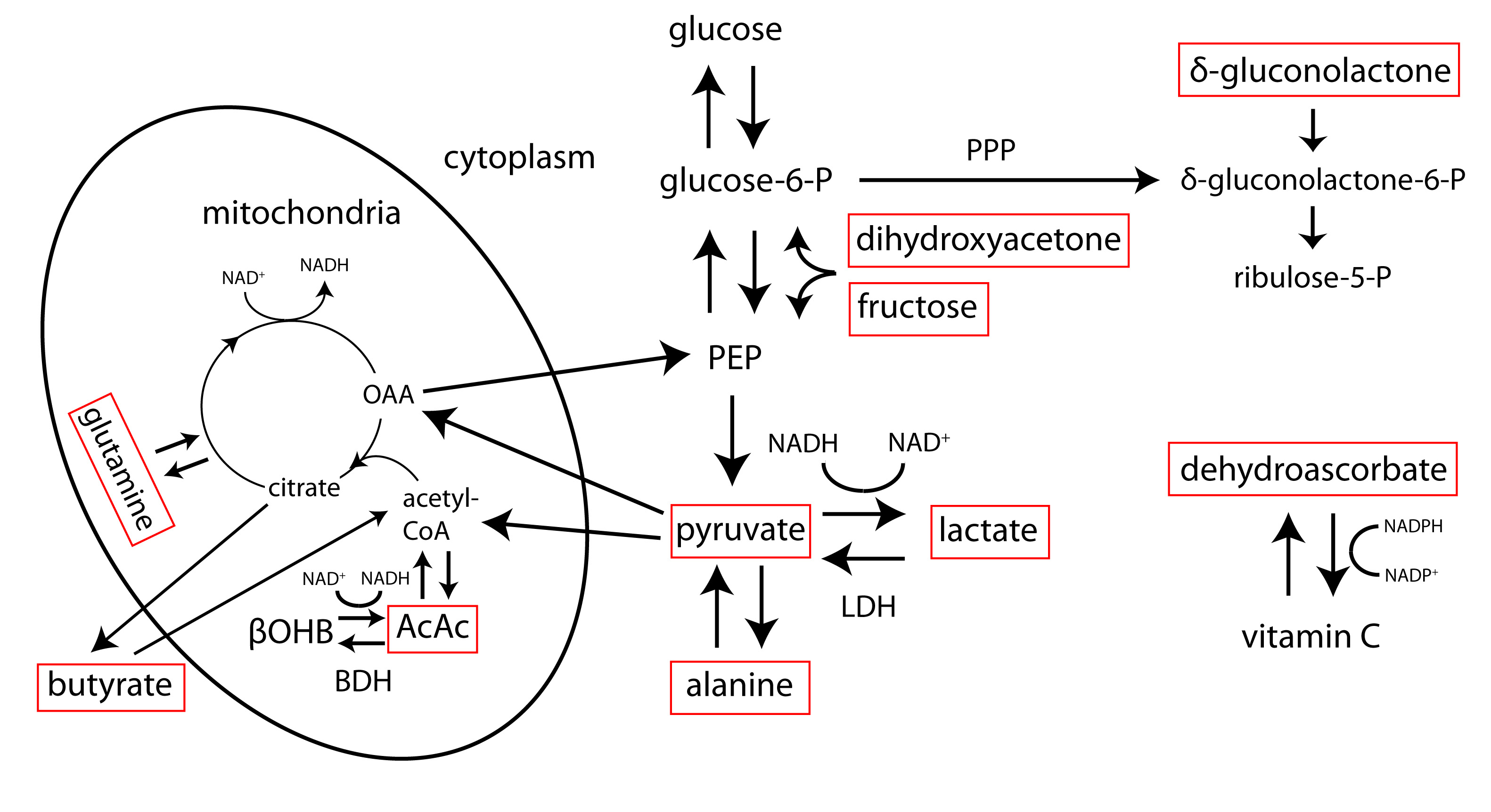
Figure 3. Simplified view of hepatic metabolic pathways accessible via hyperpolarized 13C MRI. Some relevant hyperpolarized 13C substrates are labeled in red boxes. AcAc= acetoacetate, βOHB= β-hydroxybutyrate, LDH= lactate dehydrogenase, BDH= β-hydroxybutyrate dehydrogenase, OAA= oxaloacetate, PEP= phosphoenolpyruvate, PPP= pentose phosphate pathway, NAD(H)= nicotinamide adenine dinucleotide (hydride), NADP(H)= nicotinamide adenine dinucleotide phosphate (hydride)
Target applications of liver HP 13C MRI#
Liver cancer
Primary liver cancer, predominantly hepatocellular carcinoma (HCC), is the fourth leading cause of cancer death worldwide, accounting for 780,000 deaths per year (41). Numerous studies have demonstrated that HP 13C MRI can provide diagnostic information in preclinical models of liver cancer, based on results with [1-13C]pyruvate and other 13C probes. Similar to other cancers, a highly consistent finding has been increased HP 13C pyruvate to lactate flux in liver tumors, driven by increased aerobic glycolysis associated with malignant transformation. Two early studies clearly demonstrated high HP lactate and alanine signals in orthotopic HCC tumors implanted into rats, correlated with LDH and ALT expression levels (11,42). Another study, as illustrated in Figure 4, also demonstrated increased HP lactate and alanine signals in a sophisticated, switchable transgenic model of liver cancer, which receded with tumor regression (43). The HP signals again correlated well with genetic signatures including the concentration of LDH and ALT as measured by ex vivo activity assays. Notably, this study identified increased HP alanine production as a very early indicator preceding tumorigenesis. Another study compared HP [1-13C]pyruvate with FDG-PET for imaging of HCC in rats (44), finding that either approach could visualize the increased glycolytic metabolism in these tumors.
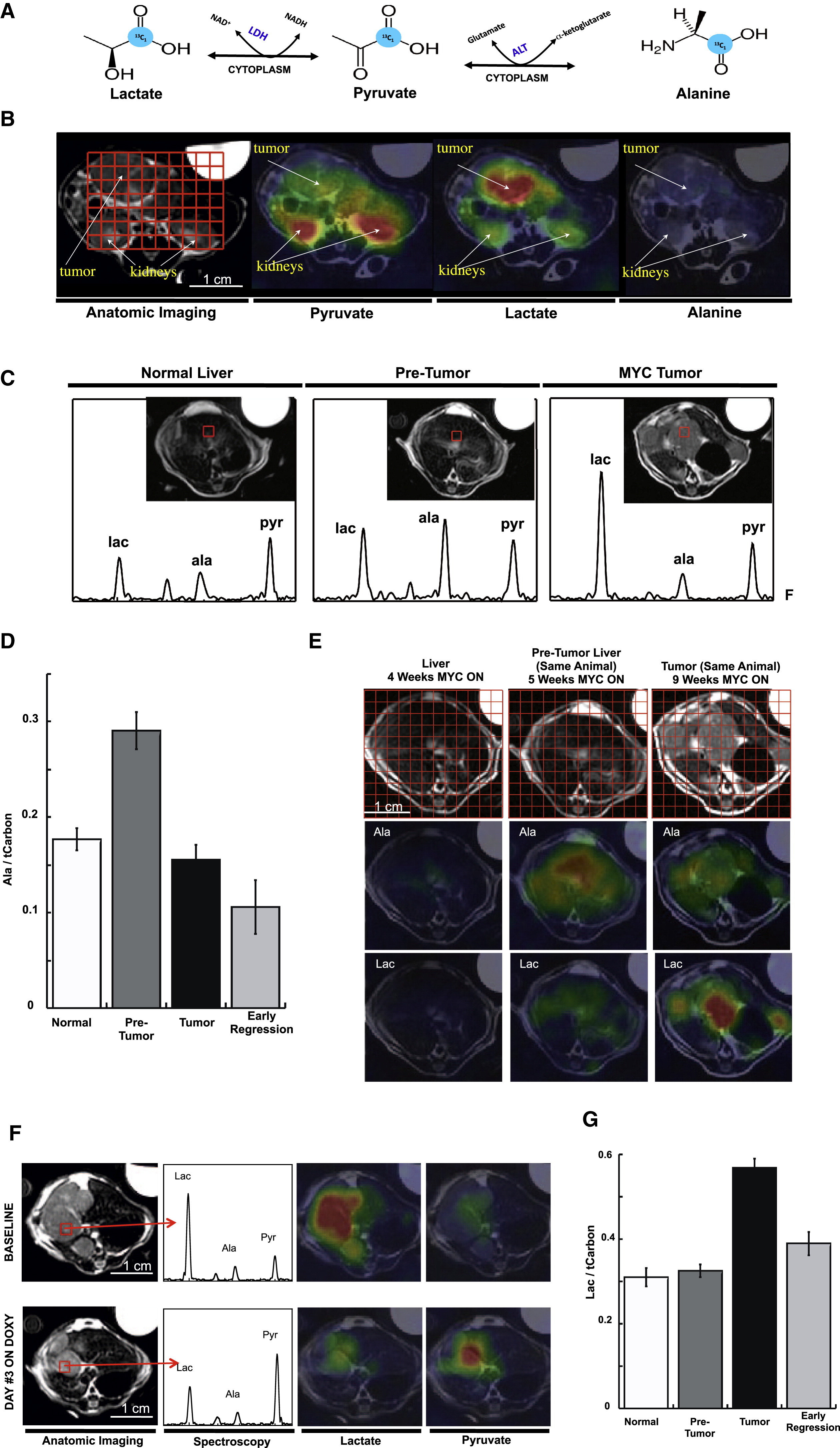
Figure 4. HP [1-13C]pyruvate MRI of a switchable, transgenic mouse model of liver cancer. A) Metabolic interconversion of HP [1-13C]pyruvate with [1-13C]lactate and [1-13C]alanine. B) Axial images of individual HP metabolites of [1-13C]pyruvate overlaid in color on conventional 1H images in grayscale, in a mouse with transgenic liver cancer. C) Evolution of metabolite signals with tumor progression, with group summaries shown in D&G. E) Heterogeneity of liver signals with progression. F) Change in liver signals with disease regression. Figure reproduced from Hu S et al. (43)
HP 13C probes other than [1-13C]pyruvate have also been described in application to imaging of liver tumors. Increased glutamine consumption is a well-recognized aberration of tumor metabolism (45), potentially providing energy, carbon skeletons, and dampening of oxidative stress. Flux of HP [5-13C]glutamine to [5-13C]glutamate has been used to detect glutaminase activity in hepatoma cells (46). These results were carried forward in vivo by another group who demonstrated the ability to detect increased [5-13C]glutamate product signal in cancerous as compared with normal liver tissue (47). [1,3-13C2]ethyl acetoacetate has been proposed as a marker for liver cancer, based on upregulated uptake and esterase activity in liver tumors (48).
Recent preclinical HP 13C MRI work on liver cancer has focused on the potential to assess treatment response non-invasively. One study comprehensively examined the potential of [1-13C]pyruvate, [13C]urea, and [1,4-13C2]fumarate to detect response of orthotopic rat HCC to transcatheter arterial embolization (TAE), demonstrated clear responses to treatment in the HP signals (49). Urea and pyruvate signals were both locally decreased in the treated tumor region, with an increased apparent rate of lactate production. Fumarate to malate conversion correlated with histologically confirmed necrosis, based on the low permeability of the dicarboxylate molecule fumarate through intact plasma membranes (50). A recent related study showed the potential for [1-13C]pyruvate MRI to detect latent domains of viable HCC surviving TAE treatment (51).
In addition to primary tumors, liver is a frequent site of metastases for many cancers, such as breast, colorectal and prostate. While little preclinical HP 13C imaging work has been conducted in the area of liver metastases, a recently published study shows proof of principle clinical translation for detecting increased 13C lactate production in human liver metastases from prostate cancer, as well as the potential for monitoring response to therapy (carboplatin + docetaxel, in this case) (52). Some results from this preliminary human translational study are shown in Figure 5.
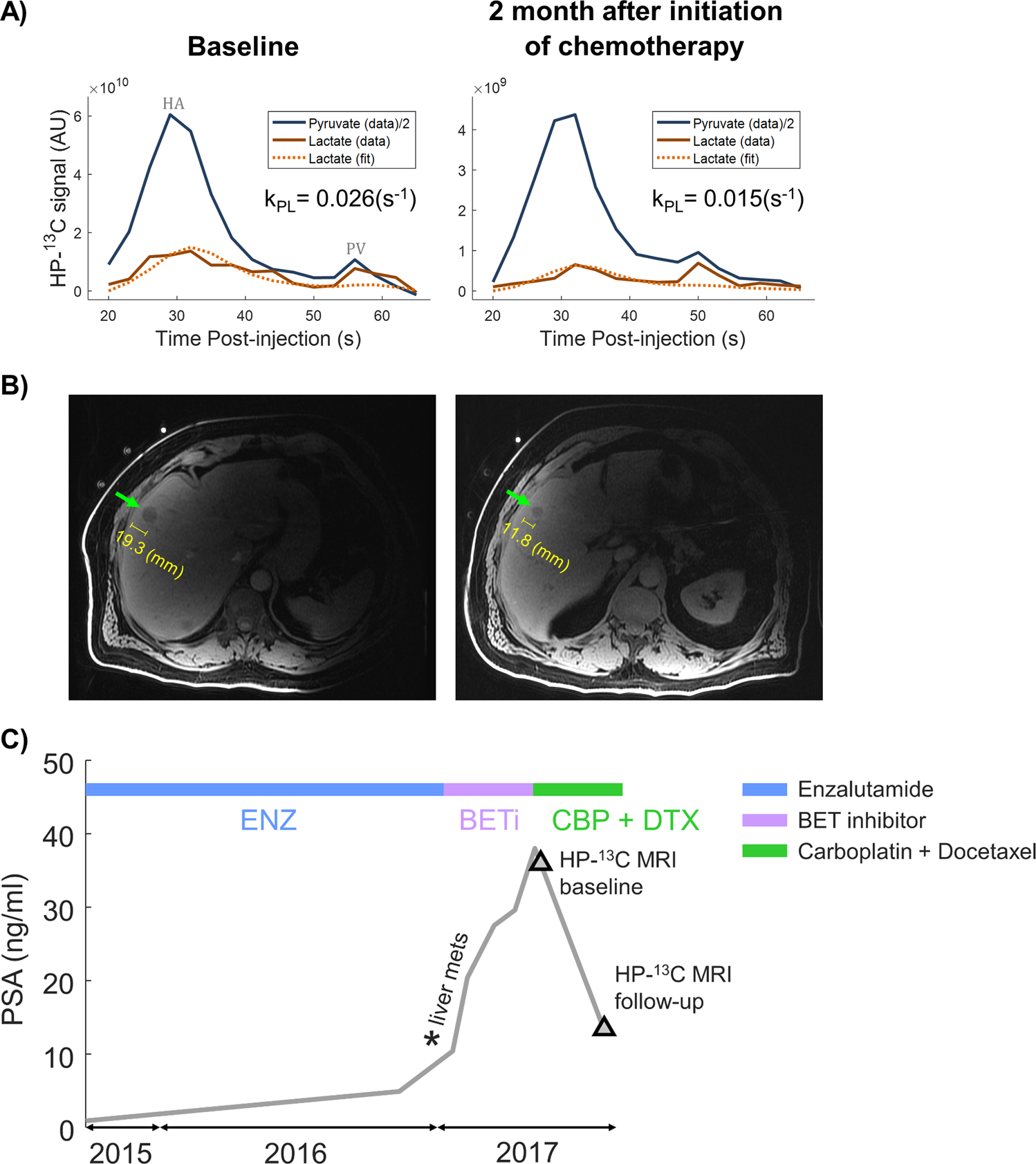
Figure 5. Proof of principle clinical translation of HP [1-13C]pyruvate MRI in a patient with liver metastases from prostate cancer. Dynamic tumor-localized HP 13C metabolite signal curves are shown in (a), before and after chemotherapy (carboplatin + docetaxel). The apparent rate of conversion of HP [1-13C]pyruvate to [1-13C]lactate declined after chemotherapy. The tumor is depicted on conventional 1H MRI in (b). As shown in (c), prostate specific antigen (PSA) levels declined with chemotherapy, also indicating therapeutic response. Figure reproduced from Chen HY et al. (52)
Diffuse liver disease
HP 13C MRI also shows great potential for application to diffuse liver diseases, especially non-alcoholic steatohepatitis (NASH), a metabolically-driven hepatic inflammatory condition. While multi-echo 1H MRI provides superb depiction of liver fat, a clinically important but difficult problem is the differentiation of NASH vs uncomplicated fatty liver. Two seminal studies showed elevated conversion of [1-13C]pyruvate to both lactate and alanine in association with acute liver injury triggered by injection of carbon tetrachloride (CCl4) and 1,3-dichloro-2-propanol (1,3-DCP) (Figure 6), respectively, in rats (53,54). These agents induce liver necrosis and inflammation. Furthermore, altered conversions of [1-13C]pyruvate have been observed in proportion to dose of hepatotoxic agent (55) and duration of high fat diet (56) in other related fatty liver models, suggesting the possibility to grade disease. Decreased hepatic conversion of [1-13C]dehydroascorbate to [1-13C]vitamin C was detected after feeding mice a diet deficient in choline and methionine, a common rodent model of NASH, as a result of increased oxidative stress (Figure 7) (57). Moreover, vitamin C levels normalized after recovery on normal diet. Hepatic conversion of [2-13C]dihydroxyacetone to [2-13C]glycerol-3-phosphate is attenuated by fructose challenge (58), suggesting applicability for monitoring hepatic energy charge, which may be altered along the progression to NASH. The fructose challenge paradigm, such as previously described for hepatic 31P MRS studies (59), could be relevant for normalization of human liver HP MRI studies.
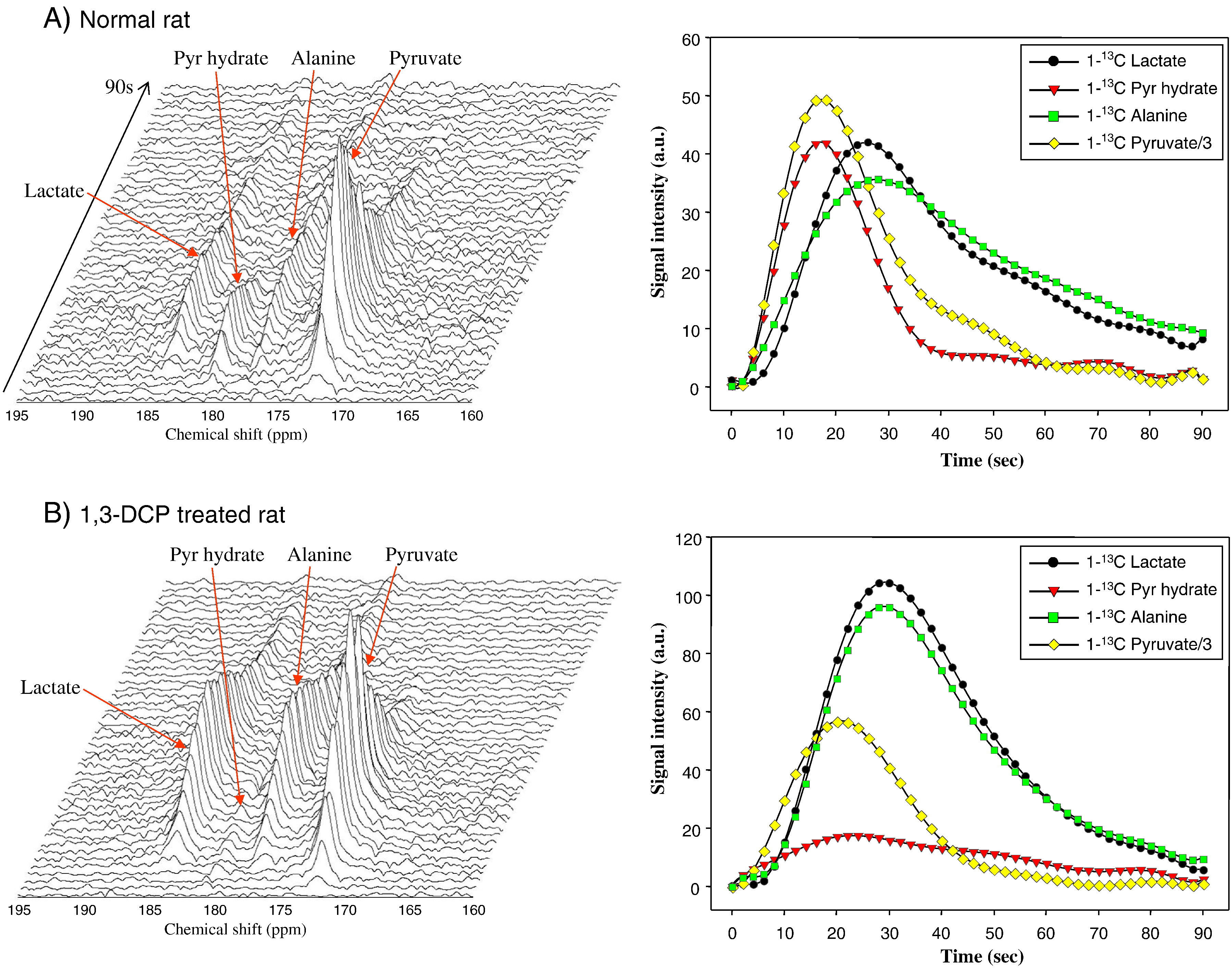
Figure 6. Example HP [1-13C]pyruvate spectra from normal rat liver (A) and rat liver from an animal treated with hepatotoxic agent 1,3-DCP (B), showing elevated conversion to [1-13C]lactate and [1-13C]alanine with treatment. Liver signal dynamics from each metabolite are shown in right column. Figure reproduced from Kim GW et al. (54)
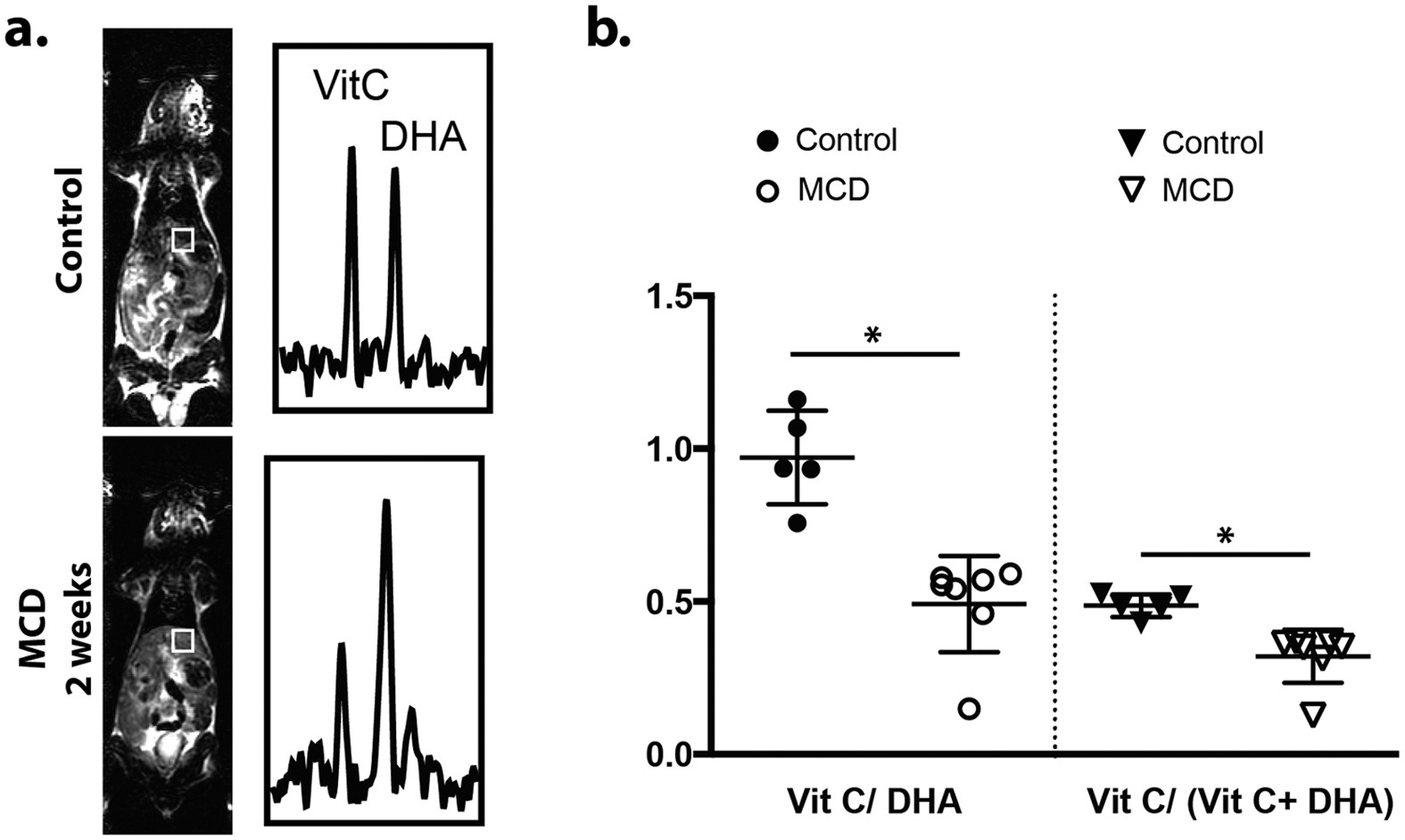
Figure 7. Example liver-localized [1-13C]dehydroascorbate spectra (a) from a normal mouse (top) and a mouse fed methionine and choline deficient diet (MCD) (bottom), a common rodent model of NASH. Summary group data are shown in (b). Production of [1-13C]vitamin C from [1-13C]dehydroascorbate is clearly attenuated in the MCD-fed group as a result of increased oxidative stress. Figure reproduced from Wilson DM et al. (57)
Ethanol has been shown to induce acute changes in HP [1-13C]pyruvate spectra in liver (60), suggesting potential applicability for studies of alcoholic fatty liver and/or alcoholic hepatitis. Altered conversions of HP [1-13C]pyruvate to lactate and alanine were also observed after ischemia-reperfusion injury in rats (61), an important complication of liver transplantation. Hepatic metabolic alterations were also detected using [1-13C]pyruvate in a model of brain death in rats (62), which also has important implications for liver transplantation.
Discussion#
Because HP 13C can target specific metabolic pathways in the liver, it offers insights into liver function that are not currently possible using other imaging techniques. In pre-clinical animal models, HP 13C MRI has demonstrated great value in assessing both liver tumors as well as diffuse liver disease such as NASH. Human liver imaging requires dealing with important technical challenges, including the dual blood flow of the liver, respiratory motion, and decreased relaxation times. Imaging with HP [1-13C]pyruvate is already feasible, and further technical developments such as improved coil arrays and the development of integrated 13C body coils are expected to make imaging more robust and reliable. Importantly HP technology allows imaging with several different targeted probes, potentially at the same time. As this exciting technology moves closer to the clinic, it will likely yield new insights into liver diseases.
References#
Luersen GF, Bhosale P, Szklaruk J. State-of-the-art cross-sectional liver imaging: beyond lesion detection and characterization. J Hepatocell Carcinoma 2015;2:101-117.
Li Q, Dhyani M, Grajo JR, Sirlin C, Samir AE. Current status of imaging in nonalcoholic fatty liver disease. World J Hepatol 2018;10(8):530-542.
Tang A, Cloutier G, Szeverenyi NM, Sirlin CB. Ultrasound Elastography and MR Elastography for Assessing Liver Fibrosis: Part 1, Principles and Techniques. AJR Am J Roentgenol 2015;205(1):22-32.
Mintorovitch J, Shamsi K. Eovist Injection and Resovist Injection: two new liver-specific contrast agents for MRI. Oncology (Williston Park) 2000;14(6 Suppl 3):37-40.
Luk WH, Au-Yeung AW, Loke TK. Imaging patterns of liver uptakes on PET scan: pearls and pitfalls. Nucl Med Rev Cent East Eur 2013;16(2):75-81.
Torizuka T, Tamaki N, Inokuma T, Magata Y, Sasayama S, Yonekura Y, Tanaka A, Yamaoka Y, Yamamoto K, Konishi J. In vivo assessment of glucose metabolism in hepatocellular carcinoma with FDG-PET. J Nucl Med 1995;36(10):1811-1817.
Roldan-Alzate A, Frydrychowicz A, Said A, Johnson KM, Francois CJ, Wieben O, Reeder SB. Impaired regulation of portal venous flow in response to a meal challenge as quantified by 4D flow MRI. J Magn Reson Imaging 2015;42(4):1009-1017.
Cunningham CH, Chen AP, Lustig M, Hargreaves BA, Lupo J, Xu D, Kurhanewicz J, Hurd RE, Pauly JM, Nelson SJ, Vigneron DB. Pulse sequence for dynamic volumetric imaging of hyperpolarized metabolic products. J Magn Reson 2008;193(1):139-146.
Gordon JW, Vigneron DB, Larson PE. Development of a symmetric echo planar imaging framework for clinical translation of rapid dynamic hyperpolarized 13 C imaging. Magn Reson Med 2017;77(2):826-832.
Miller JJ, Lau AZ, Teh I, Schneider JE, Kinchesh P, Smart S, Ball V, Sibson NR, Tyler DJ. Robust and high resolution hyperpolarized metabolic imaging of the rat heart at 7 T with 3D spectral-spatial EPI. Magn Reson Med 2016;75(4):1515-1524.
Yen YF, Le Roux P, Mayer D, King R, Spielman D, Tropp J, Pauly KB, Pfefferbaum A, Vasanawala S, Hurd R. T-2 relaxation times of C-13 metabolites in a rat hepatocellular carcinoma model measured in vivo using C-13-MRS of hyperpolarized [1-C-13]pyruvate. Nmr Biomed 2010;23(4):414-423.
Roemer PB, Edelstein WA, Hayes CE, Souza SP, Mueller OM. The NMR phased array. Magnetic resonance in medicine : official journal of the Society of Magnetic Resonance in Medicine / Society of Magnetic Resonance in Medicine 1990;16(2):192-225.
Griswold MA, Jakob PM, Heidemann RM, Nittka M, Jellus V, Wang J, Kiefer B, Haase A. Generalized autocalibrating partially parallel acquisitions (GRAPPA). Magnetic resonance in medicine : official journal of the Society of Magnetic Resonance in Medicine / Society of Magnetic Resonance in Medicine 2002;47(6):1202-1210.
Pruessmann KP, Weiger M, Scheidegger MB, Boesiger P. SENSE: sensitivity encoding for fast MRI. Magnetic resonance in medicine : official journal of the Society of Magnetic Resonance in Medicine / Society of Magnetic Resonance in Medicine 1999;42(5):952-962.
Sodickson DK, Griswold MA, Jakob PM. SMASH imaging. Magn Reson Imaging Clin N Am 1999;7(2):237-254, vii-viii.
Ohliger MA, Gordon JW, Carvajal L, Larson PEZ, Ou JJ, Agarwal S, Zhu Z, Vigneron DB, von Morze C. (55) Mn-based fiducial markers for rapid and automated RF coil localization for hyperpolarized (13) C MRI. Magn Reson Med 2021;85(1):518-530.
Shin PJ, Larson PE, Ohliger MA, Elad M, Pauly JM, Vigneron DB, Lustig M. Calibrationless parallel imaging reconstruction based on structured low-rank matrix completion. Magn Reson Med 2014;72(4):959-970.
Tropp J, Lupo JM, Chen A, Calderon P, McCune D, Grafendorfer T, Ozturk-Isik E, Larson PE, Hu S, Yen YF, Robb F, Bok R, Schulte R, Xu D, Hurd R, Vigneron D, Nelson S. Multi-channel metabolic imaging, with SENSE reconstruction, of hyperpolarized [1-(13)C] pyruvate in a live rat at 3.0 tesla on a clinical MR scanner. Journal of magnetic resonance 2011;208(1):171-177.
Boskamp E, Xie Z, Taracila V, Stephen A, Edwards M, Skloss T, Hurd R, Robb F, Murphy-Boesch J. A Dual-Tuned 70 cm Whole-Body Resonator for 13C and Proton MRI/MRS at 3T. #1714. Proc Intl Soc Mag Reson Med 26 2018.
von Morze C, Reed GD, Larson PE, Mammoli D, Chen AP, Tropp J, Van Criekinge M, Ohliger MA, Kurhanewicz J, Vigneron DB, Merritt ME. In vivo hyperpolarization transfer in a clinical MRI scanner. Magn Reson Med 2018;80(2):480-487.
Wang J, Kreis F, Wright AJ, Hesketh RL, Levitt MH, Brindle KM. Dynamic 1 H imaging of hyperpolarized [1-13 C]lactate in vivo using a reverse INEPT experiment. Magn Reson Med 2017.
Merritt ME, Harrison C, Sherry AD, Malloy CR, Burgess SC. Flux through hepatic pyruvate carboxylase and phosphoenolpyruvate carboxykinase detected by hyperpolarized 13C magnetic resonance. P Natl Acad Sci USA 2011;108(47):19084-19089.
Hu S, Chen AP, Zierhut ML, Bok R, Yen YF, Schroeder MA, Hurd RE, Nelson SJ, Kurhanewicz J, Vigneron DB. In vivo carbon-13 dynamic MRS and MRSI of normal and fasted rat liver with hyperpolarized 13C-pyruvate. Mol Imaging Biol 2009;11(6):399-407.
von Morze C, Allu PKR, Chang GY, Marco-Rius I, Milshteyn E, Wang ZJ, Ohliger MA, Gleason CE, Kurhanewicz J, Vigneron DB, Pearce D. Non-invasive detection of divergent metabolic signals in insulin deficiency vs. insulin resistance in vivo. Sci Rep 2018;8(1):2088.
von Morze C, Chang GY, Larson PE, Shang H, Allu PK, Bok RA, Crane JC, Olson MP, Tan CT, Marco-Rius I, Nelson SJ, Kurhanewicz J, Pearce D, Vigneron DB. Detection of localized changes in the metabolism of hyperpolarized gluconeogenic precursors (13) C-lactate and (13) C-pyruvate in kidney and liver. Magn Reson Med 2017;77(4):1429-1437.
Lewis AJ, Miller JJ, McCallum C, Rider OJ, Neubauer S, Heather LC, Tyler DJ. Assessment of Metformin-Induced Changes in Cardiac and Hepatic Redox State Using Hyperpolarized[1-13C]Pyruvate. Diabetes 2016;65(12):3544-3551.
Hu S, Yoshihara HA, Bok R, Zhou J, Zhu M, Kurhanewicz J, Vigneron DB. Use of hyperpolarized [1-13C]pyruvate and [2-13C]pyruvate to probe the effects of the anticancer agent dichloroacetate on mitochondrial metabolism in vivo in the normal rat. Magnetic resonance imaging 2012;30(10):1367-1372.
Bastiaansen JA, Merritt ME, Comment A. Measuring changes in substrate utilization in the myocardium in response to fasting using hyperpolarized [1-(13)C]butyrate and [1-(13)C]pyruvate. Sci Rep 2016;6:25573.
von Morze C, Ohliger MA, Marco-Rius I, Wilson DM, Flavell RR, Pearce D, Vigneron DB, Kurhanewicz J, Wang ZJ. Direct assessment of renal mitochondrial redox state using hyperpolarized (13) C-acetoacetate. Magn Reson Med 2018;79(4):1862-1869.
Moreno KX, Satapati S, DeBerardinis RJ, Burgess SC, Malloy CR, Merritt ME. Real-time detection of hepatic gluconeogenic and glycogenolytic states using hyperpolarized [2-13C]dihydroxyacetone. J Biol Chem 2014;289(52):35859-35867.
Keshari KR, Wilson DM, Chen AP, Bok R, Larson PE, Hu S, Van Criekinge M, Macdonald JM, Vigneron DB, Kurhanewicz J. Hyperpolarized [2-13C]-fructose: a hemiketal DNP substrate for in vivo metabolic imaging. J Am Chem Soc 2009;131(48):17591-17596.
Marco-Rius I, Wright AJ, Hu DE, Savic D, Miller JJ, Timm KN, Tyler D, Brindle KM, Comment A. Probing hepatic metabolism of [2-(13)C]dihydroxyacetone in vivo with (1)H-decoupled hyperpolarized (13)C-MR. MAGMA 2020.
Keshari KR, Kurhanewicz J, Bok R, Larson PE, Vigneron DB, Wilson DM. Hyperpolarized 13C dehydroascorbate as an endogenous redox sensor for in vivo metabolic imaging. Proc Natl Acad Sci U S A 2011;108(46):18606-18611.
Hu S, Zhu M, Yoshihara HA, Wilson DM, Keshari KR, Shin P, Reed G, von Morze C, Bok R, Larson PE, Kurhanewicz J, Vigneron DB. In vivo measurement of normal rat intracellular pyruvate and lactate levels after injection of hyperpolarized [1-(13)C]alanine. Magnetic resonance imaging 2011;29(8):1035-1040.
Park JM, Khemtong C, Liu SC, Hurd RE, Spielman DM. In vivo assessment of intracellular redox state in rat liver using hyperpolarized [1-(13) C]Alanine. Magn Reson Med 2017;77(5):1741-1748.
Chen J, Hackett EP, Singh J, Kovacs Z, Park JM. Simultaneous Assessment of Intracellular and Extracellular pH Using Hyperpolarized [1-(13)C]Alanine Ethyl Ester. Anal Chem 2020;92(17):11681-11686.
Marco-Rius I, Gordon JW, Mattis AN, Bok R, Delos Santos R, Sukumar S, Larson PEZ, Vigneron DB, Ohliger MA. Diffusion-weighted imaging of hyperpolarized [(13) C]urea in mouse liver. J Magn Reson Imaging 2018;47(1):141-151.
von Morze C, Larson PE, Hu S, Keshari K, Wilson DM, Ardenkjaer-Larsen JH, Goga A, Bok R, Kurhanewicz J, Vigneron DB. Imaging of blood flow using hyperpolarized [(13)C]urea in preclinical cancer models. Journal of magnetic resonance imaging : JMRI 2011;33(3):692-697.
von Morze C, Engelbach JA, Reed GD, Chen AP, Quirk JD, Blazey T, Mahar R, Malloy CR, Garbow JR, Merritt ME. (15) N-carnitine, a novel endogenous hyperpolarized MRI probe with long signal lifetime. Magn Reson Med 2020.
Ohliger MA, von Morze C, Marco-Rius I, Gordon J, Larson PEZ, Bok R, Chen HY, Kurhanewicz J, Vigneron D. Combining hyperpolarized (13) C MRI with a liver-specific gadolinium contrast agent for selective assessment of hepatocyte metabolism. Magn Reson Med 2017;77(6):2356-2363.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68(6):394-424.
Darpolor MM, Yen YF, Chua MS, Xing L, Clarke-Katzenberg RH, Shi W, Mayer D, Josan S, Hurd RE, Pfefferbaum A, Senadheera L, So S, Hofmann LV, Glazer GM, Spielman DM. In vivo MRSI of hyperpolarized [1-(13)C]pyruvate metabolism in rat hepatocellular carcinoma. Nmr Biomed 2011;24(5):506-513.
Hu S, Balakrishnan A, Bok RA, Anderton B, Larson PE, Nelson SJ, Kurhanewicz J, Vigneron DB, Goga A. 13C-pyruvate imaging reveals alterations in glycolysis that precede c-Myc-induced tumor formation and regression. Cell Metab 2011;14(1):131-142.
Menzel MI, Farrell EV, Janich MA, Khegai O, Wiesinger F, Nekolla S, Otto AM, Haase A, Schulte RF, Schwaiger M. Multimodal assessment of in vivo metabolism with hyperpolarized [1-13C]MR spectroscopy and 18F-FDG PET imaging in hepatocellular carcinoma tumor-bearing rats. J Nucl Med 2013;54(7):1113-1119.
DeBerardinis RJ, Cheng T. Q’s next: the diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 2010;29(3):313-324.
Gallagher FA, Kettunen MI, Day SE, Lerche M, Brindle KM. 13C MR spectroscopy measurements of glutaminase activity in human hepatocellular carcinoma cells using hyperpolarized 13C-labeled glutamine. Magn Reson Med 2008;60(2):253-257.
Cabella C, Karlsson M, Canape C, Catanzaro G, Colombo Serra S, Miragoli L, Poggi L, Uggeri F, Venturi L, Jensen PR, Lerche MH, Tedoldi F. In vivo and in vitro liver cancer metabolism observed with hyperpolarized [5-(13)C]glutamine. J Magn Reson 2013;232:45-52.
Jensen PR, Serra SC, Miragoli L, Karlsson M, Cabella C, Poggi L, Venturi L, Tedoldi F, Lerche MH. Hyperpolarized [1,3-13C2 ]ethyl acetoacetate is a novel diagnostic metabolic marker of liver cancer. Int J Cancer 2015;136(4):E117-126.
Duwel S, Durst M, Gringeri CV, Kosanke Y, Gross C, Janich MA, Haase A, Glaser SJ, Schwaiger M, Schulte RF, Braren R, Menzel MI. Multiparametric human hepatocellular carcinoma characterization and therapy response evaluation by hyperpolarized (13) C MRSI. Nmr Biomed 2016;29(7):952-960.
Gallagher FA, Kettunen MI, Hu DE, Jensen PR, Zandt RI, Karlsson M, Gisselsson A, Nelson SK, Witney TH, Bohndiek SE, Hansson G, Peitersen T, Lerche MH, Brindle KM. Production of hyperpolarized [1,4-13C2]malate from [1,4-13C2]fumarate is a marker of cell necrosis and treatment response in tumors. P Natl Acad Sci USA 2009;106(47):19801-19806.
Perkons NR, Kiefer RM, Noji MC, Pourfathi M, Ackerman D, Siddiqui S, Tischfield D, Profka E, Johnson O, Pickup S, Mancuso A, Pantel A, Denburg MR, Nadolski GJ, Hunt SJ, Furth EE, Kadlecek S, Gade TPF. Hyperpolarized Metabolic Imaging Detects Latent Hepatocellular Carcinoma Domains Surviving Locoregional Therapy. Hepatology 2020;72(1):140-154.
Chen HY, Aggarwal R, Bok RA, Ohliger MA, Zhu Z, Lee P, Gordon JW, van Criekinge M, Carvajal L, Slater JB, Larson PEZ, Small EJ, Kurhanewicz J, Vigneron DB. Hyperpolarized (13)C-pyruvate MRI detects real-time metabolic flux in prostate cancer metastases to bone and liver: a clinical feasibility study. Prostate Cancer Prostatic Dis 2020;23(2):269-276.
Josan S, Billingsley K, Orduna J, Park JM, Luong R, Yu L, Hurd R, Pfefferbaum A, Spielman D, Mayer D. Assessing inflammatory liver injury in an acute CCl4 model using dynamic 3D metabolic imaging of hyperpolarized [1-(13)C]pyruvate. Nmr Biomed 2015;28(12):1671-1677.
Kim GW, Oh CH, Kim JC, Yoon W, Jeong YY, Kim YH, Kim JK, Park JG, Kang HK, Jeong GW. Noninvasive biomarkers for acute hepatotoxicity induced by 1,3-dichloro-2-propanol: hyperpolarized 13C dynamic MR spectroscopy. Magn Reson Imaging 2016;34(2):159-165.
Moon CM, Shin SS, Heo SH, Lim HS, Moon MJ, Surendran SP, Kim GE, Park IW, Jeong YY. Metabolic Changes in Different Stages of Liver Fibrosis: In vivo Hyperpolarized (13)C MR Spectroscopy and Metabolic Imaging. Mol Imaging Biol 2019;21(5):842-851.
Moon CM, Oh CH, Ahn KY, Yang JS, Kim JY, Shin SS, Lim HS, Heo SH, Seon HJ, Kim JW, Jeong GW. Metabolic biomarkers for non-alcoholic fatty liver disease induced by high-fat diet: In vivo magnetic resonance spectroscopy of hyperpolarized [1-(13)C] pyruvate. Biochem Biophys Res Commun 2017;482(1):112-119.
Wilson DM, Di Gialleonardo V, Wang ZJ, Carroll V, Von Morze C, Taylor A, Sai V, VanCriekinge M, Bok R, Ohliger MA, Keshari KR. Hyperpolarized (13)C Spectroscopic Evaluation of Oxidative Stress in a Rodent Model of Steatohepatitis. Sci Rep 2017;7:46014.
Marco-Rius I, von Morze C, Sriram R, Cao P, Chang GY, Milshteyn E, Bok RA, Ohliger MA, Pearce D, Kurhanewicz J, Larson PE, Vigneron DB, Merritt M. Monitoring acute metabolic changes in the liver and kidneys induced by fructose and glucose using hyperpolarized [2-(13) C]dihydroxyacetone. Magn Reson Med 2017;77(1):65-73.
Abdelmalek MF, Lazo M, Horska A, Bonekamp S, Lipkin EW, Balasubramanyam A, Bantle JP, Johnson RJ, Diehl AM, Clark JM, Fatty Liver Subgroup of Look ARG. Higher dietary fructose is associated with impaired hepatic adenosine triphosphate homeostasis in obese individuals with type 2 diabetes. Hepatology 2012;56(3):952-960.
Spielman DM, Mayer D, Yen YF, Tropp J, Hurd RE, Pfefferbaum A. In vivo measurement of ethanol metabolism in the rat liver using magnetic resonance spectroscopy of hyperpolarized [1-13C]pyruvate. Magn Reson Med 2009;62(2):307-313.
Moon CM, Shin SS, Lim NY, Kim SK, Kang YJ, Kim HO, Lee SJ, Beak BH, Kim YH, Jeong GW. Metabolic alterations in a rat model of hepatic ischaemia reperfusion injury: In vivo hyperpolarized (13) C MRS and metabolic imaging. Liver Int 2018;38(6):1117-1127.
van Erp AC, Qi H, Jespersen NR, Hjortbak MV, Ottens PJ, Wiersema-Buist J, Norregaard R, Pedersen M, Laustsen C, Leuvenink HGD, Jespersen B. Organ-specific metabolic profiles of the liver and kidney during brain death and afterwards during normothermic machine perfusion of the kidney. Am J Transplant 2020;20(9):2425-2436.