HP Experimental Methods: Cells and Animals#
Renuka Sriram
Radiology and Biomedical Imaging, University of California San Francisco, San Francisco, CA 94158-2330
Corresponding author: Renuka.Sriram@ucsf.edu
Abstract
This chapter describes the essential components of pre-clinical testing of hyperpolarized 13C labeled substrates for the uninitiated. Its focus is on the unique aspects of executing a successful hyperpolarized study, starting from the dissolution stage to the delivery into the biological system. A detailed discussion of the range of model systems at various testing phases and its unique advantages and disadvantages are presented. After reading this chapter the reader should have a basic knowledge of how hyperpolarized 13C MRI probes are tested and used to investigate biomedical questions.
Keywords
Metabolism, Bioreactor, Enzyme, model systems
Chapter Content
Introduction
Dissolution – what’s in it?
Transfer to the magnet – How fast can you run?
Delivery – How much and how to?
Preclinical model systems for testing of hyperpolarized 13C probes
Understanding and interpreting the hyperpolarized signals to shed light on the underlying biochemistry of the pathology.
Further Study
References
Figure Captions
Introduction#
The development of dissolution dynamic nuclear polarization (DNP) using 13C labeled organic substrates provides significant new insights into previously inaccessible aspects of biochemical reactions. This technique of hyperpolarized (HP) 13C magnetic resonance (MR) heralds the era of metabolic imaging using MR with the added advantage of preserving the biochemical information. Over 100 13C labeled biomolecules have been hyperpolarized and tested in pre-clinical studies. These HP probes can be used to study basic biochemical reactions mediated by dehydrogenases, transaminases, decarboxylases, peptidases, acetyltransferases, acylases, kinases and hydratases, to name a few. Due both to its excellent HP MR properties and being at metabolic cross-roads, HP [1-13C]pyruvate has been extensively studied pre-clinically, across numerous pathologies and is the first HP probe used in patients (1).
This chapter lays out the essential workflow of a HP experiment, the components, pre-requisites and limitations, starting from the time the fluid comes out of the polarizer till it is injected and measured for a biomedical application. This is broken down into three steps: dissolution, transfer process to the magnet and delivery into the biological model system (Figure 1). These three steps are optimized with a keen focus on preserving the polarization. The second part of the chapter gives detailed descriptions of the variety of preclinical model systems used in the testing of HP agents.

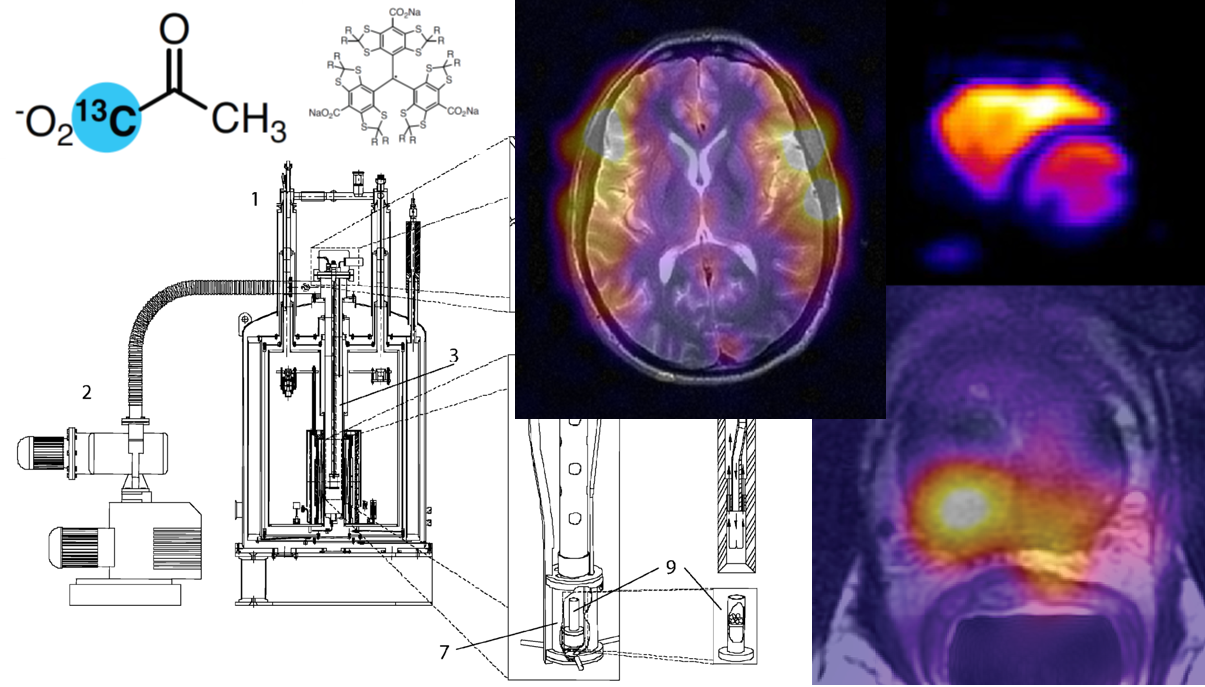
Figure 1 Key elements for preserving polarization from dissolution to delivery into biological system. This illustration shows the three major steps and the factors that determine the robustness of the HP signal. At the first stage of dissolution, its composition such as chelating agent, radical scavenger, solvent used and neutralization need to be optimized. At the subsequent step of physically transporting the polarized agent to the “measurement” magnet, a handheld electromagnet (2) or magnetic tunnel (3) to maintain low field vastly improves the HP signal lifetime. And finally, the manner of delivery and choice of model systems is crucial for successful measurement of HP agent metabolism.
Dissolution – What’s in it?#
In dDNP, the hyperpolarized substrate in the solid state is rapidly dissolved by a super-heated solution in order to create a liquid agent that has retained the hyperpolarization (See Chapter 2 for a complete description of this process). The super-heated solution is usually a buffer that has ethylenedinitrotetraacetic acid (EDTA) and a neutralizing agent. EDTA is essential to chelate paramagnetic species which would otherwise cause a loss in polarization. A neutralization agent is necessary depending on how the HP agent is prepared. For example, the workhorse of this field, [1-13C]pyruvate is prepared for polarization as a neat acid mixed with OX063 radical. Upon dissolution this would result in an acidic solution, so, in order to neutralize it the dissolution buffer usually contains equivalents of the base, sodium hydroxide. Furthermore, it has been shown that scavenging the free radical prevents loss of polarization not only at high fields but also during the transfer into the measuring magnet(4,5). However, the method of scavenging the free radicals is dependent upon the radical that has been used in the formulation of the HP agent. Although it is deemed essential to remove the radical before injection into humans, it is for the large part not removed in preclinical experiments. That being said one may want to consider it seriously in the case of HP agents with short relaxation times.
With the development of varied HP agents having low relaxation times or polarizations to study diverse metabolic pathways, optimizations to minimize the loss of polarization is centered around the use of deuterated solvents (in addition to deuterating the HP agent itself, see chapter 6) in the dissolution. Labile protons would then be exchanged with the deuterons, resulting in lesser dipolar relaxation of the 13C nucleus (as much as two bonds away) and thereby extending the lifetime of the HP agent. A recent publication by Cho et al (6) has shown an increase from 20% for HP glutamine to two fold for HP urea upon the use of deuterated solvents during dissolution.
Transfer to the magnet – How fast can you run?#
Once dissolved, the HP agent decays with the spin–lattice relaxation time \(T_{1}\), which is on the order of 10s of seconds. Invariably, the polarized solution needs to be transferred from the polarizer to the “measurement” magnet, which could result in dramatic losses of polarization since the \(T_{1}\) is field-dependent. It would be pertinent to be cognizant of the stray magnetic fields in the vicinity and along the pathway of the polarizer and the “measurement” magnet. This can be measured using a three axis Hall probe. Most established labs have a “marked” or “known pathway that needs to be followed when carrying the polarized dissolution solution to the magnet for measurement. Some recent developments for this express purpose of facilitated transfer has been the use of constant low field via magnetic tunnels (3) or carriers (2). Given the exponential nature of the time constant, at 3 times the \(T_{1}\) only 5% of the signal remains. Hence, to enable rapid transfer of the polarized solution directly into the animal or scanner, mechanized pneumatic systems have also been developed (Fig. 2.1) (7-10). This would prove invaluable in cases of HP probes with very short \(T_{1}\)’s. And the simplest alternative, in most labs, due to lack of magnetic tunnel or mechanized direct line infusion pumps, is usually a “runner” who efficiently draws up the solution from the polarizer after dissolution, after thorough mixing, and hands off to another person standing by the sample into which it would be injected.
Delivery – How much and how to?#
The penultimate step before acquisition of signal is delivery of the HP agent into the biological system of interest. And the two main aspects of this step are: i) how much of the agent to deliver and ii) the mechanism of delivery. Although these two aspects have a wide range of solutions depending on the biological model system chosen, some of the key facets to bear in mind are discussed here with further details pertinent to each type of model discussed in the next section.
How much of the hyperpolarized agent is sufficient?#
While the native concentration of the HP agent is dictated by the formulation for polarization, it is further diluted twice. Once upon dissolution and then again subsequently upon injection into solutions with cells/tissue or via the blood pool in an intact animal before metabolism of the agent is observed. While the amount or concentration of the HP agent needs to be optimized when going from an in vitro to an in vivo model system, the key factors that are to be considered in both are
a) Sufficient delivery of agent to ensure that the failure to observe metabolism of probe is not due to inadequate signal strength. This can be achieved by titrating from a large concentration of HP agent to a lesser concentration so as to observe a constant robust signal of the metabolic product. It is imperative to use the minimal concentration of the probe so as not to alter the equilibrium of the system under study. Since, unlike PET (concentration of probes are in nanomoles), the concentrations of HP agents are usually in the range of 100s of mM.
b) Consider loss of signal from HP agent due to nonspecific binding to macromolecules. For example, when testing in cells, the presence of albumin in the serum added to the media of cell culture causes a signal loss of ~10% of the HP agent. Thus, serum free media is usually used for testing HP agents in intact cells.
How to deliver the hyperpolarized agent?#
Upon thorough mixing of the dissolution care must be taken to not have any air bubbles while drawing up the solution into a syringe. The presence of bubbles has two major implications: 1) destruction of the MR signal due to susceptibility artifacts caused by inhomogeneous magnetic fields and 2) air bubbles can be lethal if injected intravenously into an animal. To minimize the presence of air bubbles, it is recommended to draw up the solution in a deliberate motion and not very swiftly. Furthermore, attempts to remove air bubbles by constantly moving the syringe plunger up and down and agitating the solution is discouraged as it would result in further loss of signal. The second aspect of the HP agent delivery is the rate of injection. Be it an in vitro or an in vivo model system, too fast an injection would result in turbulent flow (in case of cells in solution) causing apparent reduced signal intensity or be physiologically detrimental in case of animals (11). Also, the order of the HP agent delivery needs to be considered. In the case of testing using cell cultures, an optimal method would be the introduction of the HP agent first into the MR tube followed by the cell containing solution. Although logistically this may not be feasible for all in vitro model systems (such as a perfusion bioreactor loop), this provides a unique advantage of a baseline of the HP agent (and its contaminant), before the cells arrive at the MR sensitive region of the coil. This often proves invaluable in cases of confounding results with regards to observation of metabolic products overlapping with contaminants or pre-existence of product of interest in the dissolution and kinetic modeling (12).
Preclinical model systems for testing of hyperpolarized 13C agents#
In developing and optimizing a HP agent, the general approach consists of the following steps (described in detail in Chapter 8). Initial in vitro chemistry studies are performed in order to maximize HP probe concentration, polarization, and \(T_{1}\) relaxation, while minimizing contaminants. Usually as part of these chemistry studies both the probe polarization and dissolution processes are optimized, and the magnetic field dependence of the \(T_{1}\) relaxation of the HP probe and low field probe transfer effects (quadrupolar and paramagnetic relaxation effects) are determined. The probe is then tested in enzyme systems to assess the detection and rate of metabolic byproducts formation, to demonstrate feasibility. The subsequent step often involves the injection of the HP agent in living cells or tissues of HP agent uptake and metabolism under controlled conditions. The final step usually involves in vivo studies to demonstrate biodistribution, probe uptake, metabolism, and overall imaging feasibility and clinical significance. The potential clinical utility of a HP agent can be established in these studies, which can be used in the FDA IND application for its eventual use in patients.
A template of preclinical model systems used for systematic testing, development and validating a HP agent for successful clinical translation is shown in Table 1. While there’s an assortment of preclinical models that one could use for the testing of HP agent, the choice is heavily dependent on the question one is trying to address. In a simplistic view, this is presented here as three phases – namely feasibility, development and validation. It is important to note here, that the in vitro and ex vivo models used in feasibility and development, usually employ high-resolution magnets with narrow bore and very simple pulse and acquire pulse sequences. Its only with the escalation of validation using in vivo animal models, imaging magnets and sophisticated spatially and frequency selective pulse sequences are employed (see chapter 3). Furthermore, these two phases of testing - namely feasibility and development are applicable to only biochemically active HP agents such as pyruvate or bicarbonate and not perfusion agents such as urea.
Table 1 Preclinical model systems for testing HP agents
Stage |
Model/System |
Salient Features |
|---|---|---|
Feasibility |
Naïve enzymes |
- Simplest system |
Cell lysates |
- Multicomponent system |
|
Development |
Intact cells in solution |
- Complete in vitro model |
Bioreactor (Perfusion system) |
- Robust, physiologically relevant conditions |
|
Bioreactor |
- Unique model system with limited intact microenvironment |
|
Bioreactor |
- Comprehensive analysis of single organ metabolism without confounding recirculation issues |
|
Validation |
In vivo animal models |
- Necessary step for clinical translation |
Models used for feasibility testing#
Once a successful HP agent has been successfully formulated and its polarization has been optimized, the two simplistic models used to evaluate the reaction kinetics, binding affinity, substrate inhibition are the use of:
a) Naïve enzyme in a tube - This entails the addition of isolated enzymes at an optimal concentration in a physiologic buffer and temperature in an MR compatible receptacle, usually a 5 or 10 mm MR tube pursuant to the RF coil available for testing, with the appropriate cofactors and ligands. Enzymes are usually found in many isoforms and derived from various animal sources, so care must be taken to use the appropriate isoform and species (to ensure homology to humans). This is a powerful methodology that can help enumerate reaction kinetics (13), detect otherwise invisible low-population intermediate species (14). In addition to enzymology, this technique has unequivocal ability to shed insights into molecular (15), protein-ligand interactions and protein folding (13).
b) Cell lysates - In case of enzymes that are cost prohibitive or simply unavailable in the purified form, use of cell lysate is a popular alternative. Roughly 50 million cells are required for one HP experiment. The cells are lysed mechanically (stator-rotor) and chemically (detergent) to ensure maximal disruption of intracellular organelle membranes when studying non-cytosolic enzymes in ice cold conditions in a buffer containing protease and phosphatase cocktail mixture to prevent degradation of proteins. The cell debris is discarded, and the soluble supernatant is then supplemented with cofactors in excess, before HP testing to account for non-availability due to nonspecific binding to other enzymes present in the lysate. An inherent drawback of using this model system for initial testing prior to an in vivo application is the lack of intact cell membrane, which obfuscates any transport concerns. That being said, this model system has proven to be invaluable to decipher that the basis of HP signal is mainly exchange driven and not net production.
Models used for HP agent development for a predetermined biomedical application#
Upon confirmation of measurable enzymatic activity of the HP agent, the next logical step would be its testing in an intact biological system. This comprises of basic cells in solution or a complex perfusion system to be used in conjunction with cells, tissues or organs.
a) Cells in solution – This is the simplest model system, attested by the number of published studies using this methodology, as it only entails an aggregate of viable cells in an NMR tube. However similar to the cell lysate experiment, this requires ~50 million cells for one replicate and the cells cannot be reused. Some of the caveats of using this system are – 1)potential alteration of the metabolic state of the cells (due to immediate measurement after cell detachment in case of adherent cells, transition from being on ice , a common practice to slow down metabolic processes, to biologically relevant temperatures all under the auspices of non-homogenous oxygen availability) and 2) longitudinal studies are not feasible in this model system as the viability of the cells is greatly reduced over time from warm ischemia, excessive concentration of HP agent and its additives. This modest system has been successfully used for extensive kinetic models (12,16) (see chapter 7), to assess the rate of transport (17), as well as to interrogate simultaneous, alternate pathways of metabolism (18).
b) Bioreactors - Bioreactors as the name suggests are reaction vessels with tightly controlled parameters of nutrient supply, oxygenation, pH, removal of waste for monitoring biochemical processes. Such closed loop reactors have been engineered as perfusion systems that provide a physiologically pertinent culture condition to study metabolism of excised organs such as heart and liver using stable carbon-13 labeled isotopes. These can be adapted to be MR compatible for HP applications. The schematic shown in figure 3, clearly details the multiple components of the cell incubator adapted for hyperpolarized NMR studies. The continuous nutrient supply is established via circulating media, maintained at 37 °C with optimal dissolved oxygen content to the cells/tissue/organ kept in place to minimize motion and prevention of removal via flow of recirculating media. This is a sophisticated complex system that is developed and implemented in individual academic labs and involves elaborate setup of plastic tubes, usually 2-6 meters long, for the circulating media, effective sterilization of the bioreactor parts in contact with cells for prolonged experiments and ensuring bubble free environment in the RF coil sensitive region for good shim homogeneity. This powerful platform has several unique advantages: 1) simultaneous multinuclear detection such as 31P for bioenergetics monitoring, 13C for combined hyperpolarized and thermal isotopic flux monitoring; 2) ability to carefully control multiple physiologic parameters such as pH, perfusion, oxygen and temperature and observe its consequence on cellular metabolism; 3) perform longitudinal studies on the same cells to assess therapeutic response and proliferative status; 4) minimized sample requirements; 5) adaptable for concurrent PET applications (19). This has been successfully used to study cell, tissue and organ metabolism in situ.
Cells: The cells are restricted within the MR sensitive region of the reservoir by numerous methods(20), each having distinct advantages. However, the cells under study heavily influences the choice of immobilization in the bioreactor tube. For instance, strongly adherent cells are studied on polystyrene beads, such as macrophages (21) or primary cells (22), while most immortalized cells do well in alginate encapsulation or hollow fibers.
Intact tissue: The use of a bioreactor to study intact tissues provides an exclusive method to study certain diseases where appropriate model systems are sorely lacking such as benign renal tumors (23). This is a nondestructive method which can preserve the precious clinical samples for further correlative biochemical and immunohistochemical assays on the same sample. However, a rather large infrastructure as well as specialized tools is required to execute this pre-clinical yet highly valuable platform to study clinically relevant disease processes. Some of the essential elements are 1) preparation of tissue (such as slices) without compromising tissue viability using specialized apparatus, 2) sterile culture of the tissues in specially formulated and tested media, optimized oxygen and media flow conditions to maintain the metabolism over time and finally 3) careful loading of the tissues into specifically designed cartridge-tissue holders for bioreactor chambers to preserve a homogenous magnetic field.
Perfused Organs: While perfused organs have been used for a long time to study intermediate metabolism in intact organs, only the perfused heart and liver has been employed for hyperpolarized NMR. This requires a very specialized skill set and is an order of magnitude more complex than cell studies in bioreactors. While the media and perfusion conditions are well worked out this requires expertise in leak proof cannulation of the aorta or portal vein (for heart and liver respectively)
(24). This has a distinct advantage over in vivo measures of these organs due to the increased sensitivity from inherent localization and high filling factor enabling the study of metabolic intermediates otherwise inaccessible for a better understanding of the biochemical pathways.
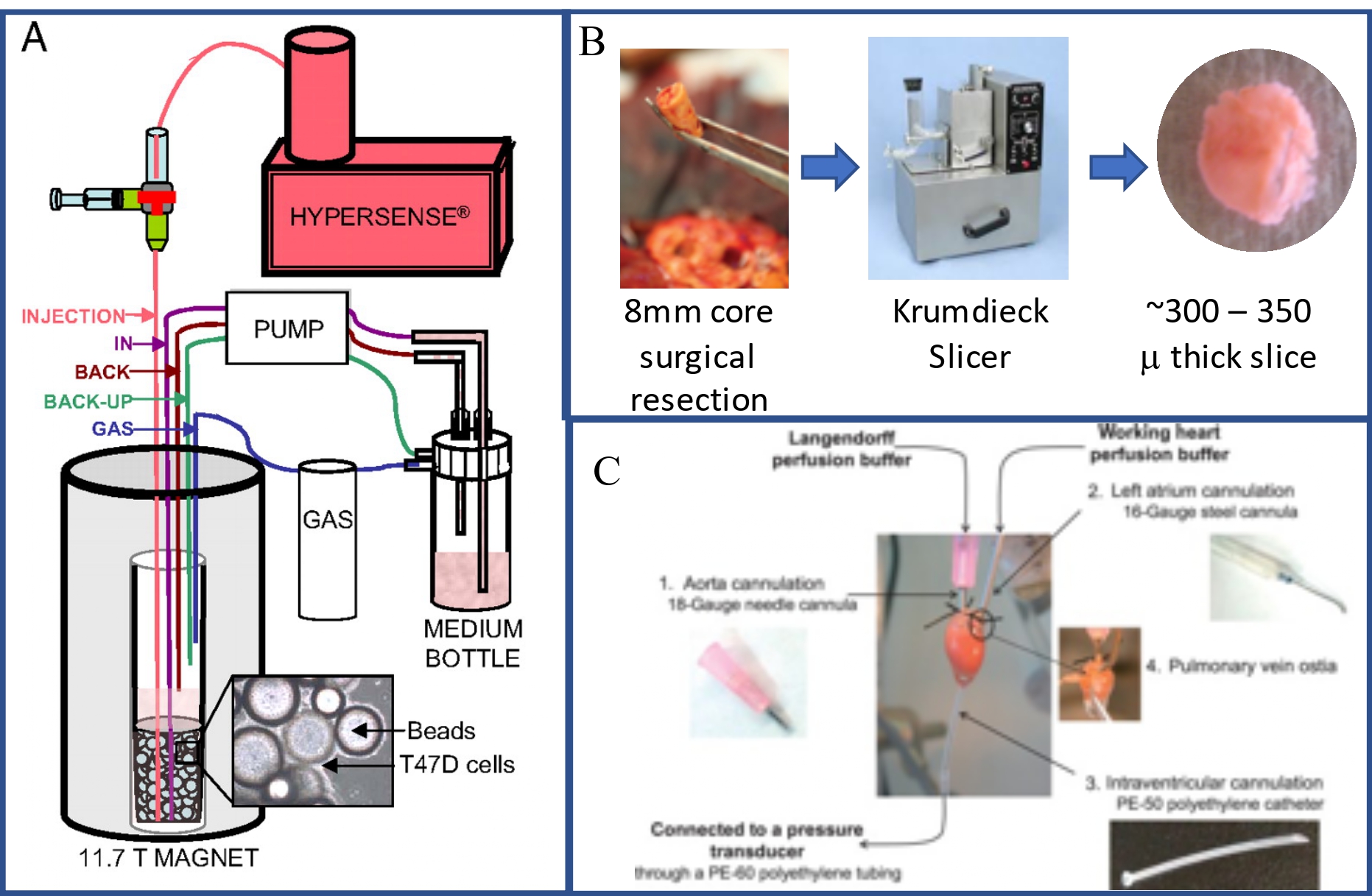
Figure 2: Components of bioreactor system to study metabolism of cells, tissues and organs. A)Adapted from Harris et al (25), this figure shows the schematic of the bioreactor. It comprises of a fluid path containing the circulating media (via a peristaltic pump). The media also has a separate gas port for oxygen dissolution into the media. The injection port is provided to infuse hyperpolarized substrate from the polarizer (Hypersense). The inset shows the cells grown on polystyrene beads. This same schema can be used for tissue and organ studies by replacing the cell/bead by tissue slices or perfused organs. B) This subfigure, shows the components required to achieve thin tissue sections for use with bioreactor. This involves, procuring a 8 mm core from surgical resection, followed by slicing into 300-350 micron thick slices using a Krumdieck slicer. C)This image shows the components of a Langendorff perfused heart, adapted from (26)
Pre-clinical animal models used in hyperpolarized NMR#
Animal models represent the penultimate step before clinical translation of any new diagnostic or therapeutic technology. While the use of large animals, especially non-human primates, is closer to human biology such as organ morphology and physiology, logistics and availability of disease models dictates the extensive use of smaller animal models like rats and mice. While the biological relevance of the choice of animal model transcends all applications, we highlight some of the considerations that are specific to its use for hyperpolarized MR studies to study biochemical processes.
Animal preparation for MR imaging#
Planning of hyperpolarized experiments on animals require some specific physiological and technical considerations.
Vascular access - Tail vein is by far the most often used route as it provides for easy repetitive cannulation in mice and rats, which in itself is a skill indispensable for longitudinal studies. A key requirement for successful HP study is the maintenance of patent vascular catheterization over prolonged time periods (1- 4 hours) for injection of hyperpolarized substrates. This involves periodic flushing of the cannula with heparinized saline to prevent clotting of the backflowing blood. In order to avoid volume-loading the animal (especially mice) over the course of a HP experiment, the catheter length should be minimized. Of note, in our experience, the saline flush following the injection of hyperpolarized agent (to avoid increased probe injection) results in a second peak of the HP signal, which often complicates the kinetic modeling. Hence, the saline flush is usually performed only after the HP experiment is completed.
Physiological status - Similar to most other imaging studies, rigorous control of functional parameters such as heart and respiration rate, body temperature is required. Additional factors that have a direct impact on the measure HP signal are:
I. Level of anesthesia – similar to other functional imaging applications, hyperpolarized signals are sensitive to the depth of anesthesia. Josan et al (27) showed that in rats, the pyruvate signal observed in the brain was dependent on the anesthetic depth. In conjunction, it has been recently shown that type of anesthetic also determines the rate of conversion (28).
II. Nutritional state – Hyperpolarized signals are acutely sensitive to the fed/fasted state of the animal. With this powerful technology, multiple pathways of metabolism can be measured simultaneously (unlike PET). For example, in the liver, pyruvate can be metabolized via multiple pathways depending on the state of the animal. It has been shown in rat livers that the pyruvate is metabolized via the PDH (pyruvate dehydrogenase) when fed, while in the fasted condition, pyruvate is catabolized via pyruvate carboxylase (29). Hence, in order to measure hyperpolarized bicarbonate signal, it is necessary to ensure that the animal is not fasted. Similarly a reduction in hyperpolarized alanine signal has been observed in rat livers (30). Careful thought also needs to be given to the hyperpolarized agent being used to interrogate a particular organ. As illustrated by Bastiaansen et al (31), in the rodent heart, hyperpolarized butyrate and pyruvate gives a measure of fatty acid oxidation and carbohydrate metabolism, respectively. While hyperpolarized butyrate transport remained unchanged between fed and fasted state, pyruvate metabolism was altered in the fasted state. Hence, to decrease any confounding factors and variability in the results between animals, it is recommended to carefully control for the fed/fasted state of the animal and the time of day the experiments are conducted.
![Effects of Anesthetic agents on rodent brain metabolism of HP [1-13C]pyruvate](../_images/CH0004_OCFigure%2B4.3_Sriram_v1_Orig.jpg)
Figure 3: Effect of different anesthetic agents on rodent brain metabolism of HP [1-13C]pyruvate. Reproduced with permission from Marjańska et al (28), this figure shows the A) 13C spectra acquired 15 s after injection of HP [1-13C]pyruvate solution in two representative rats under isoflurane and morphine. B) Representative in vivo time courses of HP bicarbonate signal as a function of the anesthetic agent is shown. Maximal bicarbonate signal is observed under morphine and pentobarbital resulted in diminished bicarbonate signal from HP [1-13C] pyruvate.
Hyperpolarized substrate dosing considerations#
Unlike PET tracers, the injected doses of hyperpolarized substrates are often much higher than the equilibrium concentrations found at normal physiology. While it has been shown that the lactate and alanine signal is not saturated when using pyruvate concentrations of up to 0.7 mmol/kg in the fed state in mouse and rat kidney, the dose dependent signals of the downstream metabolites are variable in the different organs for different metabolites. For instance in the rodent cardiac metabolism, pyruvate concentrations greater than 0.4mmol/kg causes saturation of bicarbonate and lactate signal while there is a linear correspondence with alanine signal (32), whereas hyperpolarized alanine and bicarbonate signals plateau above 0.2 mmol/kg in the rodent liver (33,34). Additionally, physical impediments of certain organs like the blood brain barrier highly influences the dose of HP agent used which is a function of its permeability.
Understanding and interpreting the hyperpolarized signals to shed#
light on the underlying biochemistry of the pathology.
Several pre-clinical works have shown that in the classical example of hyperpolarized pyruvate conversion to lactate, the signal is a function of not only the enzyme activity and the availability of NADH cofactor but also dependent on the product pool sizes (35-37). These findings highlight the multiple components regulating the hyperpolarized signal and the complexities of the underlying enzymatic reaction. While under in vitro conditions, it is feasible to estimate the absolute flux of the hyperpolarized probes in conjunction with thermal stable isotope resolved metabolomics or radioisotope studies, under in vivo conditions, it becomes convolved with other effects such as signal contamination from vascular component, uncertainty in the concentration of the hyperpolarized probe in the tissue of interest, intra- and extra-cellular compartmentalization, and the effect on the hyperpolarized signal with unknown and varying levels of enzyme and cofactor. This also implies that to thoroughly understand the enzyme system under study using hyperpolarized MR techniques, it is imperative to do extensive pre-clinical studies in order to fully utilize this powerful technique to uncover kinetic mechanisms in real time.
ACKNOWLEDGEMENTS
We would like to acknowledge the funding sources P41 EB013598 (NIH) and PC160630 (DoD) and the members of the Hyperpolarized MRI Technology Resource Center and the Pre-Clinical MR Imaging and Spectroscopy Core.
FURTHER STUDY
Bottomley, P. A., & Griffiths, J. R. (2016). Handbook of magnetic resonance spectroscopy in vivo: MRS theory, practice and applications.
References:#
1. Nelson SJ, Kurhanewicz J, Vigneron DB, et al. Metabolic Imaging of Patients with Prostate Cancer Using Hyperpolarized [1-13C]Pyruvate. Sci Transl Med 2013;5:198ra108. doi: 10.1126/scitranslmed.3006070.
2. Shang H, Skloss T, Morze von C, Carvajal L, Van Criekinge M, Milshteyn E, Larson PEZ, Hurd RE, Vigneron DB. Handheld electromagnet carrier for transfer of hyperpolarized carbon‐13 samples. Magn Reson Med 2016;75:917–922. doi: 10.1002/mrm.25657.
3. Milani J, Vuichoud B, Bornet A, Miéville P, Mottier R, Jannin S, Bodenhausen G. A magnetic tunnel to shelter hyperpolarized fluids. Review of Scientific Instruments 2015;86:024101. doi: 10.1063/1.4908196.
4. Miéville P, Jannin S, Bodenhausen G. Relaxometry of insensitive nuclei: Optimizing dissolution dynamic nuclear polarization. Journal of Magnetic Resonance 2011;210:137–140. doi: 10.1016/j.jmr.2011.02.006.
5. Miéville P, Ahuja P, Sarkar R, et al. Scavenging free radicals to preserve enhancement and extend relaxation times in NMR using dynamic nuclear polarization. Angew. Chem. Int. Ed. Engl. 2010;49:6182–6185. doi: 10.1002/anie.201000934.
6. Cho A, Eskandari R, Miloushev VZ, Keshari KR. A Non-Synthetic Approach to Extending the Lifetime of Hyperpolarized Molecules using D2O Solvation. Journal of Magnetic Resonance 2018. doi: 10.1016/j.jmr.2018.08.001.
7. Can E, Mishkovsky M, Yoshihara HAI, Kunz N, Couturier D-L, Petrausch U, Doucey M-A, Comment A. Noninvasive rapid detection of metabolic adaptation in activated human T lymphocytes by hyperpolarized 13 C magnetic resonance. Scientific Reports 2020;10:1–8. doi: 10.1038/s41598-019-57026-1.
8. Cheng T, Mishkovsky M, Bastiaansen JAM, Ouari O, Hautle P, Tordo P, van den Brandt B, Comment A. Automated transfer and injection of hyperpolarized molecules with polarization measurement prior to in vivoNMR. NMR Biomed. 2013;26:n/a–n/a. doi: 10.1002/nbm.2993.
9. Bowen S, Hilty C. Rapid sample injection for hyperpolarized NMR spectroscopy. Phys Chem Chem Phys 2010;12:5766–5770. doi: 10.1039/c002316g.
10. Bowen S, Hilty C. Time-resolved dynamic nuclear polarization enhanced NMR spectroscopy. Angew. Chem. Int. Ed. Engl. 2008;47:5235–5237. doi: 10.1002/anie.200801492.
11. Turner PV, Brabb T, Pekow C, Vasbinder MA. Administration of substances to laboratory animals: routes of administration and factors to consider. Journal of the American Association for Laboratory Animal Science : JAALAS 2011;50:600–613.
12. Harrison C, Yang C, Jindal A, DeBerardinis RJ, Hooshyar MA, Merritt M, Dean Sherry A, Malloy CR. Comparison of kinetic models for analysis of pyruvate-to-lactate exchange by hyperpolarized 13C NMR. NMR Biomed. 2012;25:1286–1294. doi: 10.1002/nbm.2801.
13. Zhang G, Hilty C. Applications of dissolution dynamic nuclear polarization in chemistry and biochemistry. Magn Reson Chem 2018;56:566–582. doi: 10.1002/mrc.4735.
14. Jensen PR, Meier S, Ardenkjaer-Larsen JH, Duus JØ, Karlsson M, Lerche MH. Detection of low-populated reaction intermediates with hyperpolarized NMR. Chem. Commun. (Camb.) 2009:5168–5170. doi: 10.1039/b910626j.
15. Lerche MH, Meier S, Jensen PR, Baumann H, Petersen BO, Karlsson M, Duus JØ, Ardenkjaer-Larsen JH. Study of molecular interactions with 13C DNP-NMR. J Magn Reson 2010;203:52–56. doi: 10.1016/j.jmr.2009.11.020.
16. Hill DK, Orton MR, Mariotti E, et al. Model free approach to kinetic analysis of real-time hyperpolarized 13C magnetic resonance spectroscopy data. PLoS ONE 2013;8:e71996. doi: 10.1371/journal.pone.0071996.
17. Reineri F, Daniele V, Cavallari E, Aime S. Assessing the transport rate of hyperpolarized pyruvate and lactate from the intra- to the extracellular space. NMR Biomed. 2016;29:1022–1027. doi: 10.1002/nbm.3562.
18. Yang C, Harrison C, Jin ES, Chuang DT, Sherry AD, Malloy CR, Merritt ME, Deberardinis RJ. Simultaneous Steady-state and Dynamic 13C NMR Can Differentiate Alternative Routes of Pyruvate Metabolism in Living Cancer Cells. Journal of Biological Chemistry 2014;289:6212–6224. doi: 10.1074/jbc.M113.543637.
19. Keshari KR, Wilson DM, Van Criekinge M, et al. Metabolic response of prostate cancer to nicotinamide phophoribosyltransferase inhibition in a hyperpolarized MR/PET compatible bioreactor. Prostate 2015;75:1601–1609. doi: 10.1002/pros.23036.
20. Penet MF, Shah T, Wildes F, Krishnamachary B, Bharti SK, Torres JP, Artemov D, Bhujwalla ZM. MRI and MRS of intact perfused cancer cell metabolism, invasion, and stromal cell interactions. NMR Biomed. 2019;32:13641. doi: 10.1002/nbm.4053.
21. Sriram R, Nguyen J, Santos JD, Nguyen L, Sun J, Vigneron S, Van Criekinge M, Kurhanewicz J, MacKenzie JD. Molecular detection of inflammation in cell models using hyperpolarized 13C-pyruvate. Theranostics 2018;8:3400–3407. doi: 10.7150/thno.24322.
22. Chaumeil MM, Radoul M, Najac C, Eriksson P, Viswanath P, Blough MD, Chesnelong C, Luchman HA, Cairncross JG, Ronen SM. Hyperpolarized 13C MR imaging detects no lactate production in mutant IDH1 gliomas: Implications for diagnosis and response monitoring. NeuroImage: Clinical 2016;12:180–189. doi: 10.1016/j.nicl.2016.06.018.
23. Sriram R, Van Criekinge M, DeLos Santos J, Keshari KR, Peehl DM, Wang ZJ. Non-Invasive Differentiation of Benign Renal Tumors from Clear Cell Renal Cell Carcinomas Using Clinically Translatable Hyperpolarized 13C Pyruvate Magnetic Resonance. Tomography 2016;2:35–42. doi: 10.18383/j.tom.2016.00106.
24. Lumata L, Yang C, Ragavan M, Carpenter N, DeBerardinis RJ, Merritt ME. Hyperpolarized (13)C Magnetic Resonance and Its Use in Metabolic Assessment of Cultured Cells and Perfused Organs. Meth. Enzymol. 2015;561:73–106. doi: 10.1016/bs.mie.2015.04.006.
25. Harris T, Eliyahu G, Frydman L, Degani H. Kinetics of hyperpolarized 13C1-pyruvate transport and metabolism in living human breast cancer cells. Proc. Natl. Acad. Sci. U.S.A. 2009;106:18131–18136. doi: 10.1073/pnas.0909049106.
26. Ruiz M, Gélinas R, Vaillant F, Lauzier B, Rosiers Des C. Metabolic Tracing Using Stable Isotope-Labeled Substrates and Mass Spectrometry in the Perfused Mouse Heart. Meth. Enzymol. 2015;561:107–147. doi: 10.1016/bs.mie.2015.06.026.
27. Josan S, Hurd R, Billingsley K, Senadheera L, Park JM, Yen Y-F, Pfefferbaum A, Spielman D, Mayer D. Effects of isoflurane anesthesia on hyperpolarized 13C metabolic measurements in rat brain. Magn Reson Med 2012:n/a–n/a. doi: 10.1002/mrm.24532.
28. Marjańska M, Shestov AA, Deelchand DK, Kittelson E, Henry P-G. Brain metabolism under different anesthetic conditions using hyperpolarized [1- 13C]pyruvate and [2- 13C]pyruvate. NMR Biomed. 2018;31:e4012. doi: 10.1002/nbm.4012.
29. Jin ES, Moreno KX, Wang JX, Fidelino L, Merritt ME, Sherry AD, Malloy CR. Metabolism of hyperpolarized [1-(13)C]pyruvate through alternate pathways in rat liver. NMR Biomed. 2016;29:466–474. doi: 10.1002/nbm.3479.
30. Hu S, Chen AP, Zierhut ML, Bok R, Yen YF, Schroeder MA, Hurd RE, Nelson SJ, Kurhanewicz J, Vigneron DB. In vivo carbon-13 dynamic MRS and MRSI of normal and fasted rat liver with hyperpolarized 13C-pyruvate. Mol Imaging Biol 2009;11:399–407. doi: 10.1007/s11307-009-0218-z.
31. Bastiaansen JAM, Merritt ME, Comment A. Measuring changes in substrate utilization in the myocardium in response to fasting using hyperpolarized [1-(13)C]butyrate and [1-(13)C]pyruvate. Scientific Reports 2016;6:25573. doi: 10.1038/srep25573.
32. Schroeder MA, Atherton HJ, Cochlin LE, Clarke K, Radda GK, Tyler DJ. The effect of hyperpolarized tracer concentration on myocardial uptake and metabolism. Magn Reson Med 2009;61:1007–1014. doi: 10.1002/mrm.21934.
33. Gómez Damián PA, Sperl JI, Janich MA, Khegai O, Wiesinger F, Glaser SJ, Haase A, Schwaiger M, Schulte RF, Menzel MI. Multisite Kinetic Modeling of (13)C Metabolic MR Using [1-(13)C]Pyruvate. Radiol Res Pract 2014;2014:871619. doi: 10.1155/2014/871619.
34. Janich MA, Menzel MI, Wiesinger F, Weidl E, Khegai O, Ardenkjaer-Larsen JH, Glaser SJ, Haase A, Schulte RF, Schwaiger M. Effects of pyruvate dose on in vivo metabolism and quantification of hyperpolarized ¹³C spectra. NMR Biomed. 2012;25:142–151. doi: 10.1002/nbm.1726.
35. Kettunen MI, Hu D-E, Witney TH, Mclaughlin R, Gallagher FA, Bohndiek SE, Day SE, Brindle KM. Magnetization transfer measurements of exchange between hyperpolarized [1-13C]pyruvate and [1-13C]lactate in a murine lymphoma. Magn Reson Med 2010;63:872–880. doi: 10.1002/mrm.22276.
36. Hurd RE, Spielman D, Josan S, Yen Y-F, Pfefferbaum A, Mayer D. Exchange-linked dissolution agents in dissolution-DNP (13) C metabolic imaging. Magn Reson Med 2012;70:936–942. doi: 10.1002/mrm.24544.
37. Kettunen MI, Kennedy BWC, Hu D-E, Brindle KM. Spin echo measurements of the extravasation and tumor cell uptake of hyperpolarized [1-(13) C]lactate and [1-(13) C]pyruvate. Magn Reson Med 2012. doi: 10.1002/mrm.24591.