Integration into Cancer Studies#
Pavithra Viswanath*
Department of Radiology and Biomedical Imaging, University of California San Francisco, San Francisco, CA 94143
*Corresponding author: Pavithra.Viswanath@ucsf.edu
Abstract
Metabolic reprogramming has emerged as a fundamental hallmark of cancer. Alterations in oncogenic signaling pathways that drive tumor initiation and maintenance also rewire tumor bioenergetics and redox. This relationship provides the unique opportunity to devise metabolic imaging strategies that can inform on tumor burden and response to therapy. Hyperpolarized 13C-magnetic resonance spectroscopy and imaging (HP 13C MRS/I) is a non-invasive method of monitoring dynamic metabolic fluxes in real time in vivo. This chapter presents a detailed discussion of oncogenic metabolic reprogramming and the various HP 13C MRS/I agents that have used to interrogate this reprogramming in preclinical tumor models. Emerging human studies that point to the promise as well as challenges associated with translating HP 13C MRS/I to the clinic are also discussed.
Keywords
Cancer metabolism, metabolic reprogramming, precision imaging, magnetic resonance spectroscopy, hyperpolarized 13C imaging, oncogenic signaling pathways
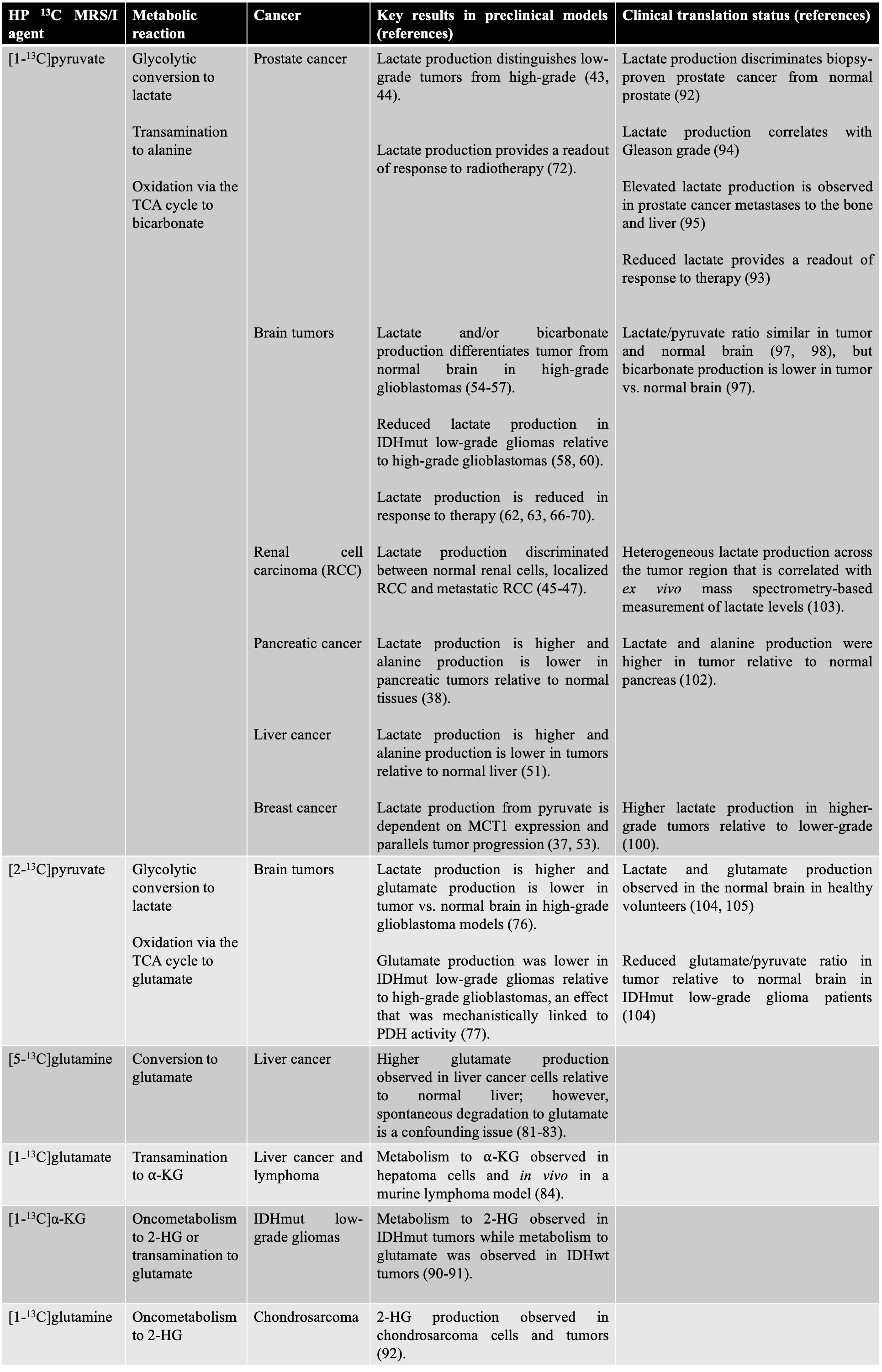
Table 1. Summary of key insights gained from preclinical and clinical studies with the various HP 13C MRS/I agents described in this chapter.
Metabolic reprogramming is a central hallmark of cancer#
Cancer is caused by genetic and epigenetic alterations in oncogenes and tumor suppressors that allow tumor cells to sustain uncontrolled proliferation, evade the immune system and invade surrounding tissue (1). Increasingly, across several tumor types, it has become clear that many of the oncogenes and tumor suppressors that drive and sustain tumorigenesis also alter metabolism in order to meet the biosynthetic, bioenergetic and redox needs of tumor cells (2-4). Specifically, oncogenic alterations allow cancer cells to access conventional and unconventional nutrient sources and alter the flux of these nutrient through metabolic pathways in order to generate biosynthetic and redox intermediates for biomass generation (2-4). Intriguingly, recent studies have begun to indicate that, beyond meeting bioenergetic needs, alterations in tumor metabolite levels can alter gene expression within the tumor as well as the surrounding tumor microenvironment (3). Collectively, these studies have established metabolic reprogramming as a fundamental cancer hallmark (1).
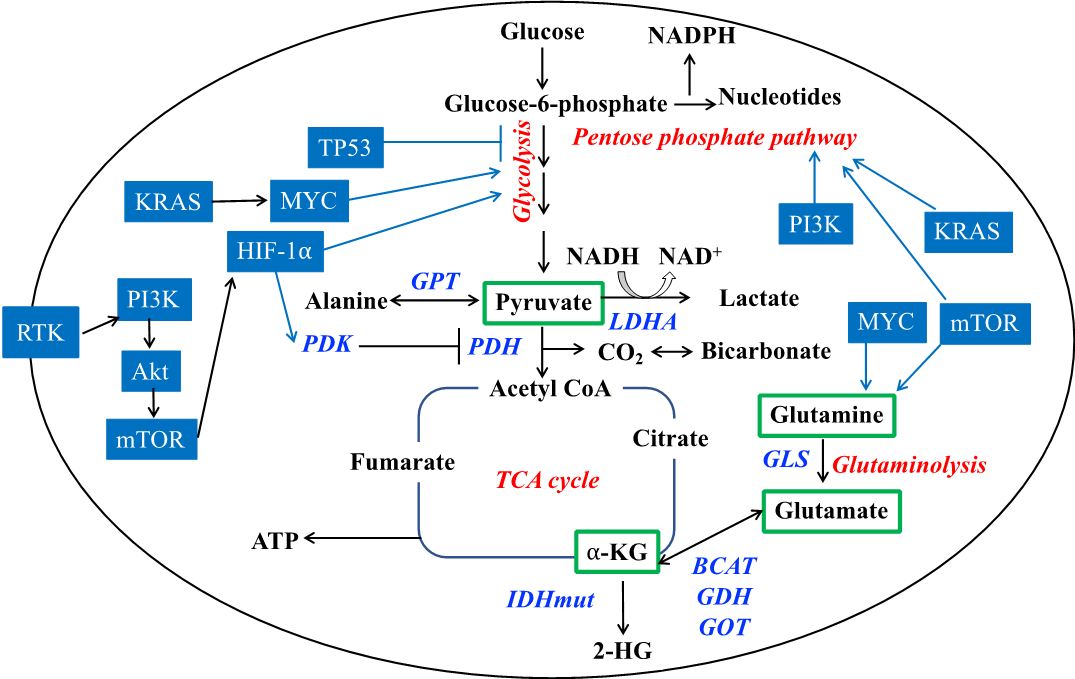
An important feature of tumor metabolic reprogramming resides in the observation that multiple oncogenic pathways converge on common metabolic phenotypes (see Figure 1) (2-4). Glucose and glutamine are the principal nutrients for mammalian cells and several oncogenic pathways, including mutations in TP53, MYC, KRAS and the PI3K-Akt-mTOR pathway, consistently reprogram glucose and glutamine metabolism (5-7). Loss of the tumor suppressor TP53, a feature of nearly 50% of all human cancers, increases glycolytic flux, alters redox balance and modulates chromatin structure via alterations in glucose-derived α-ketoglutarate (α-KG) levels (2-6). Oncogenic KRAS increases expression of the glucose transporter GLUT1 and concomitant glucose uptake and metabolism. KRAS also increases anaplerotic utilization of glutamine for biosynthesis and redox homeostasis. MYC elicits multiple metabolic effects including enhanced glycolysis, glutaminolysis, nucleotide biosynthesis and mitochondrial biogenesis. The PI3K-Akt-mTOR pathway, which is also one of the most commonly mutated pathways in cancer, links growth factor signaling to nutrient availability and ribosome biogenesis, thereby modulating glucose and amino acid utilization as well as protein translation (2-7).
 #
#
Figure 1. Oncogenic reprogramming of metabolism can be observed non-invasively with HP 13C MRS/I. Metabolism of glucose and glutamine, which are the principal cellular nutrients, can be regulated by many oncogenes and tumor suppressors including TP53, KRAS, MYC, receptor tyrosine kinases, the PI3K-Akt-mTOR pathway and the IDH1 mutation. Key oncogenes and tumor suppressors are highlighted in blue boxes. Metabolic pathways are highlighted in red. Blue arrows indicate regulation of a metabolic pathway by an oncogenic factor. Metabolic enzymes catalyzing key reactions are highlighted in blue italics. The HP 13C MRS/I probes discussed in this chapter are enclosed in green boxes. Abbreviations: BCAT: branched chain amino acid transferase; GDH: glutamate dehydrogenase; GLS: glutaminase; GPT: alanine aminotransferase; PDH: pyruvate dehydrogenase; PDK: pyruvate dehydrogenase kinase; LDHA: lactate dehydrogenase A;
The relatively recent discovery of mutations in metabolic enzymes that lead to the production of oncometabolites, i.e. metabolites that drive tumorigenesis, further highlights the central role of metabolism in cancer (6-11). The best example so far is the occurrence of mutations in isocitrate dehydrogenase (IDH) 1/2 that lead to the abnormal reduction of α-KG to 2-hydroxyglutarate (2-HG) (8-11). 2-HG inhibits the function of several α-KG-dependent dioxygenases within the cell including prolyl hydroxylases, DNA methylases, and histone demethylases, leading to an altered epigenetic state and, consequently, altered gene expression (9-11). Collectively, these alterations are now believed to kickstart tumorigenesis in 70-90% of low-grade gliomas and ~20% of acute myeloid leukemia. Other metabolic driver mutations that have been discovered in cancer are mutations in fumarate hydratase and succinate dehydrogenase, that lead to intracellular accumulation of fumarate or succinate, respectively (6).
Metabolic reprogramming can be monitored to provide precision imaging of cancer#
Non-invasive magnetic resonance imaging (MRI) is an integral part of both preclinical cancer research and clinical cancer patient management (12,13). MRI methods such as T1-weighted MRI pre- and post-contrast imaging with gadolinium-based agents, T2-weighted MRI, diffusion-weighted MRI and dynamic contrast-enhanced MRI provide exquisite anatomical detail as well as physiological information on tissue microstructure. As such, these MRI methods are indispensable for diagnosis, staging and treatment response monitoring in cancer (12-14). However, structural and physiological changes do not provide a readout of molecular events controlling tumor proliferation. In this context, there could be a significant lapse in time, which is detrimental to patient outcome, before MRI methods provide a readout of response to therapy, especially with molecularly targeted cancer therapies (15,16). Furthermore, volumetric alterations are insufficient for treatment response monitoring in the case of cytostatic tumor therapeutics. Therefore, there is a need to identify non-invasive imaging modalities that provide a readout of molecular events linked to tumor growth and proliferation (12,14,16). As described in section 8.1, metabolic reprogramming is linked to tumor bioenergetics and proliferation (1,7). This relationship provides a unique opportunity to devise non-invasive metabolic imaging strategies that can inform on tumor burden and response to therapy.
Metabolic imaging, including HP 13C MRS/I, holds great promise for precise, personalized imaging of cancer patients. In an era of precision medicine, tumor diagnosis and treatment are moving towards a paradigm characterized by analysis of individual patient genomes (pretreatment genomic analysis) such that individualized treatments tailored to the specific mutational pattern can be deployed (17,18). An important component of such precision medicine is the identification of companion diagnostics or biomarkers that will provide an early readout of treatment response (12,18,19). Since oncogenic events also drive tumor metabolic reprogramming, metabolic imaging has the potential to provide a non-invasive readout of underlying tumor genomics and aid in tumor diagnosis and/or prognosis. This is especially promising for tumors such as brain tumors, where detection of circulating tumor DNA or tumor cells does not inform on the presence of a tumor (19,20). In addition, once the patient is treated with a precision medicine method, HP 13C MRS/I has the ability to provide an early readout of response to therapy (see section 8.3.1.2), an approach which is important for longitudinal studies of response to therapy, especially for tumors that are anatomically inaccessible such as brain tumors.
Imaging oncogenic reprogramming of metabolism using HP 13C MRS/I in preclinical cancer models#
The best example of the use of cancer metabolism for non-invasive imaging is PET (positron emission tomography) imaging using 2-[18F]-fluoro-2-deoxy-D-glucose (FDG), which measures the increase in glucose uptake that is part of the Warburg effect and is observed near universally in cancer (21,22). FDG-PET imaging is used in the clinic for tumor staging and treatment response monitoring in nearly every type of cancer, with the exception of gliomas and prostate cancer (21,22). However, FDG-PET imaging as well as other PET tracers typically provide a readout of tracer uptake, but not of subsequent metabolism, thereby limiting information gain. FDG-PET can also fail to distinguish between tumor and non-cancerous processes where glucose uptake is enhanced including infection and inflammation (23). Another concern is that the radioactive nature of PET tracers prohibits repetitive longitudinal studies and limits application to children and women of reproductive age (22,23).
MRS is a complementary method of non-invasively assessing metabolic alterations (24,25). 1H-MRS informs on steady-state metabolite levels and is in clinical use (24,25). 13C MRS following administration of 13C-labeled isotopes can be performed without hyperpolarization and provides information on the dynamic flow of carbon through metabolic pathways. However, the low natural abundance of the 13C moiety (1.1%) combined with the low gyromagnetic ratio of 13C, limits the signal to noise ratio (SNR) of 13C MRS/I, leading to prohibitively long acquisition times and hampering its clinical utility (26,27). HP 13C MRS/I overcomes these issues and allows visualization of metabolic reactions in real time with high SNR (28,29). For details of DNP physics, hardware, acquisition schemes, experimental methods and data analysis, readers are encouraged to refer to chapters 1-7 in this book. The sections that follow will describe the ways in which HP 13C MRS/I agents have been used to interrogate oncogenic reprogramming of metabolism and, thereby, obtain a readout of tumor burden and treatment response in preclinical cancer models (please refer to summary in Table 1).
Interrogating the Warburg effect using HP [1-13C]pyruvate#
Aerobic glycolysis, also known as the Warburg effect, is a nearly universal metabolic hallmark of cancer (2,7,21). It describes the propensity of tumor cells to enhance glucose uptake and convert glucose almost exclusively to lactate, even under conditions of normal oxygen tension (21,30). Concomitantly, metabolism of glucose via the tricarboxylic acid (TCA) cycle is down-regulated. Relative to oxidative phosphorylation, glucose metabolism via the Warburg effect yields ~18-fold lower adenosine triphosphate (ATP) per glucose molecule. Nevertheless, despite reduced ATP production, the prevailing hypothesis (21,30) suggests that the Warburg effect benefits tumor cells in several ways. First, enhanced glycolytic flux generates precursors for various biosynthetic pathways including ribonucleotide synthesis, hexosamine biosynthesis and folate metabolism. Pyruvate conversion to lactate via the Warburg effect also maintains redox balance by regenerating NAD+ that serves as a coenzyme for redox reactions. In addition, emerging evidence indicates that the lactate secreted by tumor cells alters immune cell function in the tumor microenvironment to promote tumor invasion (31).
Several oncogenes and tumor suppressors regulate the Warburg effect in cancer (7,21,30). The PI3K-Akt-mTOR pathway and HIF-1α are notable regulators of this metabolic phenotype. Via Akt, the PI3K-Akt-mTOR pathway increases expression of GLUT1 and enhances glucose uptake (2). Constitutive stabilization of HIF-1α in many tumor types also directs glucose metabolism via glycolysis vs. the TCA cycle (32). HIF-1α increases expression of GLUT1 and lactate dehydrogenase A (LDHA), the enzyme that converts pyruvate to lactate. Concomitantly, HIF-1α upregulates expression of pyruvate dehydrogenase kinase (PDK), which mediates inhibitory phosphorylation of pyruvate dehydrogenase (PDH), the enzyme that directs decarboxylation of pyruvate to acetyl CoA. The net effect is enhanced flux of glucose-derived pyruvate to lactate and reduced pyruvate flux via the TCA cycle (2,7,32).
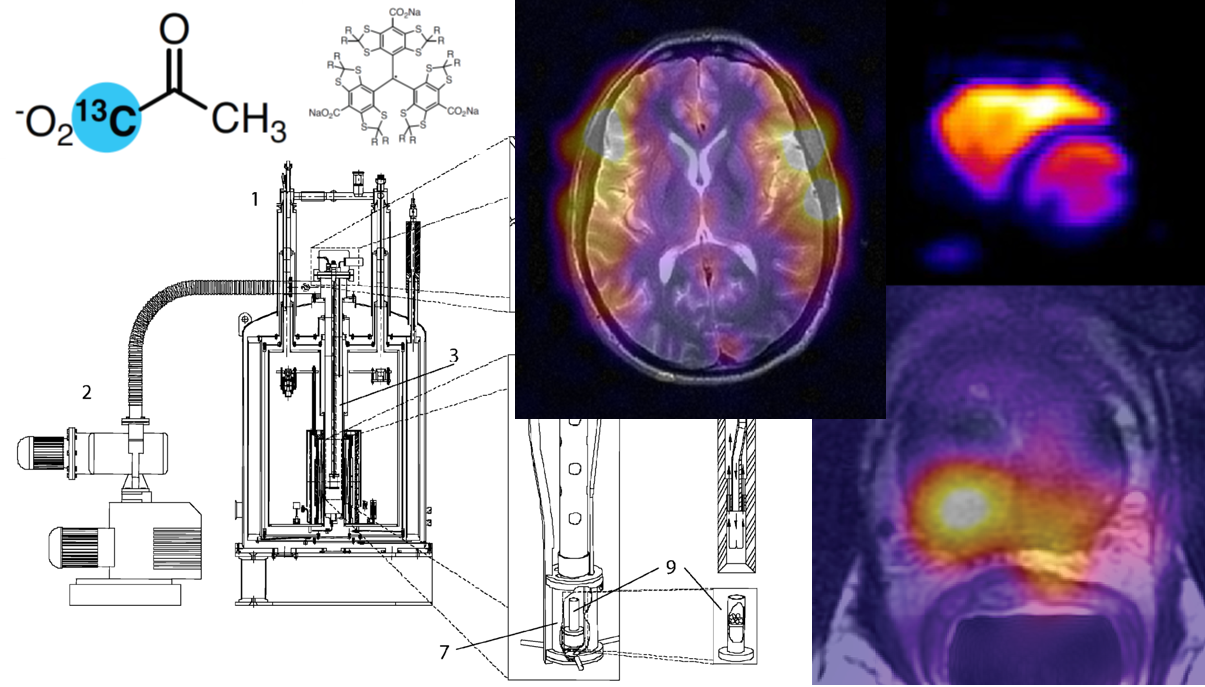
Given the widespread focus on the Warburg effect in cancer, it is not surprising that HP [1-13C]pyruvate has been the most widely used HP 13C agent in cancer (33,34). HP [1-13C]-pyruvate fulfills all the requirements for a successful HP 13C imaging agent. Pyruvate has a long T1 (~60s at the clinically relevant field strength of 3T) and polarizes well (40-50%) (34). Pyruvate transport into the cell is mediated via the monocarboxylate transporters MCT1 and MCT4, which are abundantly expressed on cells and known to mediate fast intracellular transport (35). Importantly, [1-13C]pyruvate lies at the crossroads of glycolysis, transamination and the TCA cycle. HP [1-13C]pyruvate conversion to [1-13C]lactate is a measure of LDHA activity (see Figure 1). Alternatively, HP [1-13C]pyruvate can be metabolized via PDH to produce 13CO2, which is in equilibrium with [1-13C]bicarbonate. The relative proportions of [1-13C]lactate and [1-13C]bicarbonate provide a readout of the Warburg effect. In addition, HP [1-13C]pyruvate can be converted to [1-13C]alanine by alanine aminotransferase. Although less well-studied than pyruvate to lactate conversion, studies have begun to indicate that alanine aminotransferase couples alanine generation from pyruvate to α-KG production from glutamate, thereby facilitating the flow of glutamine-derived carbon into the TCA cycle (36).
Multiple factors influence HP [1-13C]pyruvate metabolism. Expression levels of the pyruvate and lactate transporters MCT1 and MCT4 determine HP [1-13C]pyruvate delivery into the cell and, in the case of brain tumors, across the blood brain barrier (BBB) (29,37). Activity of LDH and alanine aminotransferase determine flux from HP [1-13C]pyruvate to lactate and alanine respectively (refer to Figure 1) (38,39). In addition, the steady-state pool sizes of lactate and alanine also play a role since the HP 13C pool is in equilibrium with the thermally polarized 13C pool (39). Another important consideration is the steady-state concentration of the co-factor for pyruvate to lactate conversion i.e. NADH and, therefore, the NAD+/NADH ratio (40). Mechanistic studies of HP [1-13C]pyruvate metabolism need to therefore account for all these factors. As new HP 13C agents are developed, similar aspects of transport, enzyme activity, co-factor dependence and steady-state metabolite concentrations need to be investigated. (See HP Agents and Biochemical Interactions for additional discussion of these various factors.)
Monitoring tumor burden, aggressiveness and disease progression#
Across tumor types, elevated [1-13C]pyruvate to lactate conversion has been shown to be capable of differentiating tumor from normal tissue and assessing tumor aggressiveness in preclinical cancer models (see summary in Table 1) (29,33).
In prostate cancer, which is one of the major causes of death in men, patients who are identified as low-risk based on histopathological Gleason scoring often harbor high-grade tumors and, therefore, there is an urgent need to identify non-invasive methods that can provide a readout of tumor aggressiveness (41,42). In the transgenic adenocarcinoma of mouse (TRAMP) prostate cancer model, HP [1-13C]lactate production from pyruvate can differentiate between normal prostate and tumors graded as low- or high-grade based on histopathology (43). In a subsequent study with patient-derived prostate tissue slices assembled into perfused bioreactor systems (see HP Experimental Methods: Cells and Animals for further details of perfused bioreactors), significantly higher conversion from HP [1-13C]pyruvate to lactate was observed in malignant tissue slices relative to benign prostate tissues (44). Mechanistically, higher pyruvate to lactate conversion was associated with enhanced expression of the transporters MCT1 and MCT4 as well as higher expression of LDHA.
HP [1-13C]pyruvate is informative of aggressiveness in preclinical renal cell carcinoma (RCC) models (45-47). Due to increased availability and utilization of MRI, incidental detection of RCCs has increased (48). However, MRI is unable to differentiate between low grade, indolent RCCs, which can be managed with active surveillance, from high grade aggressive RCCs that require surgery (48). HP lactate production as well as lactate efflux discriminated between normal renal epithelial cells, localized RCC and metastatic RCC, thereby providing a readout of tumor aggressiveness (46). These results were correlated with expression levels of MCT4. In addition to HP pyruvate-to-lactate conversion, assessment of intracellular and extracellular 13C lactate pools via diffusion-weighted HP 13C MRS/I in vivo correlated with tumor LDHA and MCT4 expression and tumor aggressiveness (45).
Pancreatic cancer is a devastating disease that is potentially curable by surgery if detected at an early stage (49,50). Therefore, new imaging biomarkers that facilitate early detection of pancreatic cancer are needed (50). Interestingly, analysis of HP [1-13C]pyruvate metabolism in genetically engineered mouse models of pancreatic ductal adenocarcinoma showed a decrease in [1-13C]alanine/[1-13C]lactate following progression from lower-grade lesions to high-grade pancreatic tumors (38). This effect was accompanied by an increase in steady-state lactate levels and LDH activity and a reduction in alanine aminotransferase activity during disease progression (38).
Similar results were reported in a genetically engineered mouse model of MYC-driven hepatocellular carcinoma (51). Like pancreatic cancer, hepatocellular carcinoma is a leading cause of cancer-related mortality and differentiation of malignant tumors from benign tumors and non-cancerous liver cirrhosis is challenging (52). HP [1-13C]pyruvate conversion to alanine was higher in in pre-tumor tissues relative to normal liver and established tumors. In contrast, HP [1-13C]pyruvate conversion to lactate increased as tumors developed and was rapidly decreased during tumor regression (51).
Breast cancer accounts for approximately a quarter of all cancer cases and is a leading cause of death among women. Like many other cancers, breast cancers reprogram their metabolism to enable tumor proliferation and display a pronounced Warburg effect. An early study of live breast cancer cells in a perfused bioreactor system showed HP [1-13C]pyruvate conversion to lactate that was mechanistically correlated with MCT1 expression (37). A subsequent study in a doxycycline-inducible MYC-driven murine breast cancer model showed lactate production from HP [1-13C]pyruvate that paralleled MYC-driven tumor induction or regression (53)`.
HP [1-13C]pyruvate to lactate can monitor tumor burden in preclinical models of high-grade primary glioblastomas (54-56). One of the earliest studies of HP [1-13C]pyruvate was conducted in rats carrying orthotopic U87-MG and U251-MG tumor xenografts and showed that the HP lactate/pyruvate ratio could differentiate between tumor and normal brain (55). A subsequent study showed that tumor regions with necrosis or hypoxia demonstrated a smaller magnitude of increase in the HP lactate/pyruvate ratio relative to normal brain when compared to tumor regions that were not necrotic/hypoxic (54). These results suggest that HP [1-13C]pyruvate has the potential to assess intratumoral heterogeneity in cellularity or oxygenation. Interestingly, a recent study suggested that HP [1-13C]lactate production from pyruvate was not higher in tumor relative to normal brain in patient-derived glioblastoma models as opposed to established glioblastoma cell lines that have been in culture for a long time (i.e. the U87 or U251 models) (56). These results point to importance of patient-derived models in preclinical tumor imaging and have important implications for the application of HP [1-13C]pyruvate to glioblastoma patients as discussed in the section on clinical applications of HP 13C MRS/I (see section 8.4).
Most studies of HP [1-13C]pyruvate have investigated conversion to lactate as a measure of the Warburg effect. However, as described above, the Warburg effect also involves concomitant down-regulation of pyruvate flux into the TCA cycle via PDH. Monitoring HP [1-13C]pyruvate conversion to HP [1-13C]bicarbonate, in addition to conversion to HP [1-13C]lactate, would enable assessment of this dual shift in metabolism towards glycolysis and away from the TCA cycle. In addition, measuring the ratio of two products i.e. the lactate/bicarbonate ratio provides a potentially more robust metric that is not dependent on substrate delivery as opposed to the ratio of product to substrate. However, the detection of HP [1-13C]bicarbonate can be challenging due to the low SNR of the signal. Assessment of HP [1-13C]pyruvate conversion to lactate and bicarbonate rats carrying orthotopic C6 glioma tumors showed that the HP lactate/bicarbonate ratio was higher in the tumor relative to normal contralateral brain. This study used a slightly higher concentration of pyruvate (125 mM as opposed to the normally used 80 mM) to achieve sufficient SNR (57).
In contrast to the high-grade primary glioblastomas and, indeed, most cancers, low-grade gliomas characterized by a mutation in isocitrate dehydrogenase 1 (IDHmut) do not display a Warburg effect (10). Glucose uptake and concomitant lactate production are reduced in IDHmut low-grade glioma cells, leading to a reduced intracellular pool of lactate (58). In addition, expression of MCT1, MCT4 and LDHA are reduced in IDHmut low-grade glioma cells, an effect linked to promoter hypermethylation-mediated silencing (58,59). Collectively, these effects lead to reduced production of HP [1-13C]lactate from HP [1-13C]pyruvate and, potentially, reduced ability to monitor tumor burden (58,60). Interestingly, a recent study suggested that lactate production from HP [1-13C]pyruvate varied among patient-derived IDHmut low-grade glioma models in a manner dependent on tumor aggressiveness (61).
Monitoring tumor response to therapy and development of treatment resistance#
HP [1-13C]pyruvate has been shown to provide a readout of response to therapy in several cancer types (Table 1) (29,33,34). Treatment response is generally linked to reduced HP [1-13C]pyruvate to lactate conversion and mediated by molecular alterations in transporters, LDH activity or cofactor concentrations described earlier. In high-grade primary glioblastomas, HP [1-13C]pyruvate has been used to monitor response to standard of care therapy i.e. radiotherapy and chemotherapy with temozolomide (62,63). HP [1-13C]pyruvate to lactate conversion is reduced in rats bearing orthotopic U87 tumors treated with temozolomide (63). Importantly, this response is observed within 24 h of exposure to the chemotherapeutic agent temozolomide (TMZ), at a timepoint that precedes MRI-detectable reduction in tumor volume, indicating that reduced HP [1-13C]lactate production from HP [1-13C]pyruvate can serve as an early indicator of response to TMZ therapy (63). Furthermore, there was no alteration in HP [1-13C] pyruvate to lactate conversion in U87 tumors expressing O6-methylguanine DNA methyltransferase, an enzyme that confers resistance to TMZ, indicating that HP [1-13C]pyruvate can also inform on resistance to therapy (63). Mechanistically, reduced HP [1-13C]lactate production following TMZ treatment was not mediated by alterations in NADH concentration or LDH activity, but rather by reduced expression and activity of the pyruvate kinase isoform PKM2, a glycolytic enzyme that indirectly controls pyruvate metabolism. In another study, treatment of rats bearing orthotopic C6 glioma xenografts with radiotherapy led to a significant reduction in lactate production from HP [1-13C]pyruvate within 96 h, even as an increase in tumor volume was observed, indicating that HP [1-13C]pyruvate has the ability to inform on response to radiotherapy (62).
HP [1-13C]pyruvate also provides a readout of response to molecular targeted therapies. As described earlier (see section 8.1), activating mutations in the PI3K-Akt-mTOR pathway are observed in most cancers, including high-grade primary glioblastomas, and often lead to activation of the Warburg effect and higher LDH activity (2,7,21,30). Multiple inhibitors of PI3K or mTOR have been identified and are in clinical trials against various tumor types (64). Treatment with these inhibitors often leads to tumor stasis rather than tumor shrinkage, thereby providing a rationale for the identification of non-invasive imaging biomarkers that can inform on tumor response to therapy (65). Examination of glioblastoma cells treated with the PI3K inhibitor LY294002 or the mTOR inhibitor everolimus pointed to a significant reduction in HP [1-13C]pyruvate conversion to lactate relative to untreated controls, an effect that was mechanistically linked to reduced LDH activity downstream of PI3K inhibition (66,67). Similar results were observed in orthotopic glioblastoma tumor xenografts treated with everolimus or the PI3K-mTOR inhibitor XL765 (68,69). Both agents induced a significant reduction in HP lactate production from pyruvate that preceded volumetric alterations.
As described above (see section 8.3.1.1), most studies of HP [1-13C]pyruvate metabolism have focused on assessing pyruvate conversion to lactate, partly due to the relatively less SNR of HP [1-13C]bicarbonate. However, a few studies have used the HP lactate/bicarbonate ratio to detect tumor burden and response to therapy (57,70). Dichloroacetate (DCA) is a pyruvate analog which inhibits PDK, thereby relieving inhibitory phosphorylation of PDH and leading to increased PDH activity (71). DCA forces tumors to shunt pyruvate away from glycolysis and towards the TCA cycle. This metabolic rewiring has been shown to inhibit GBM growth (71). A study in rats carrying orthotopic C6 glioblastoma tumors showed that the HP lactate/bicarbonate ratio was higher in the tumor relative to normal contralateral brain (41,57). Treatment with DCA restored the lactate/bicarbonate ratio in the tumor to levels seen in normal brain, pointing to the utility of the HP lactate/bicarbonate ratio in assessing response to DCA in glioblastomas (57). Another study examined the response of C6 glioblastomas to anti-angiogenic therapy targeting the vascular endothelial growth factor (VEGF) (70). In a manner similar to DCA, there was a rapid reduction in the tumor lactate/bicarbonate ratio to levels approaching normal contralateral brain following a single dose of anti-VEGF antibody, indicating that HP [1-13C]pyruvate conversion to lactate and bicarbonate can provide a readout of anti-angiogenic therapy.
In the TRAMP model of prostate cancer, HP [1-13C]pyruvate was able to inform on tumor response to radiotherapy (72). Serial imaging showed significant reductions in HP pyruvate to lactate conversion in tumors responding to radiotherapy, with concomitant significant increases in HP pyruvate to alanine conversion. Furthermore, alterations in HP lactate production were dependent on radiation dose with higher radiation doses producing larger reductions in HP lactate production relative to lower radiation doses. These effects were associated with corresponding decreases in LDH activity (72). Similar results were observed in another study focused on squamous cell carcinoma and colon cancer tumors in vivo, wherein reduced HP [1-13C]pyruvate conversion to lactate was observed following radiotherapy, an effect also associated with reduced LDH activity (73). Interestingly, the lactate/pyruvate ratio was significantly lower following irradiation of colon tumor xenografts, which were relatively less hypoxic, while the reduction in the lactate/pyruvate ratio was relatively modest in the more hypoxic squamous cell carcinoma tumors (73). These results again serve to highlight the potential of HP 13C MRS/I in assessing tumor variability in hypoxia.
As evidence accumulates for the utility of HP [1-13C]pyruvate in monitoring treatment response, it becomes important to define those applications where HP 13C MRS/I can yield information that cannot be obtained by other metabolic imaging modalities, particularly FDG PET imaging, which has been widely used in several tumor types to measure early response to therapy. In this context, a recent study compared the ability of FDG-PET and HP [1-13C]pyruvate to detect altered glycolytic metabolism accompanying cell death induced by a TRAIL agonist (MEDI3039) in colorectal (Colo205) and breast adenocarcinoma (MDA-MB-231) xenografts in mice (74). HP [1-13C]pyruvate conversion to lactate was reduced following treatment and preceded alterations in tumor volume. Mechanistically, this effect was associated with reduced glucose flux to lactate, presumably resulting in reduced steady-state lactate concentration. In contrast, FDG uptake and phosphorylation were not altered following treatment, suggesting that HP [1-13C]pyruvate metabolism is a more sensitive marker of early treatment-induced alterations in glycolytic flux than FDG PET imaging (74).
Interrogating glycolysis and the TCA cycle using HP [2-13C]pyruvate#
HP [2-13C]pyruvate differs from [1-13C]pyruvate in the positional labeling of the carbon backbone of pyruvate. HP [2-13C]pyruvate has the advantage of simultaneously providing a measure of glycolytic and TCA cycle flux. Via glycolysis, [2-13C]pyruvate is converted to [2-13C]lactate. When [2-13C]pyruvate is shunted via the TCA cycle, the 13C label is observed in [5-13C]glutamate and, in some cases, [1‐13C]citrate and [5-13C]α‐KG (75-77). The rate-limiting enzyme in this pathway is PDH and, therefore, HP [2-13C]pyruvate provides a readout of PDH activity (77,78). In addition, conversion of pyruvate-derived acetyl CoA to [1‐13C]acetylcarnitine and [1‐13C]acetoacetate also provides a measure of the activity of enzymes involved in fatty acid and ketone body metabolism. HP [2-13C]-pyruvate, therefore, has the ability to non-invasively report on several metabolic reactions i.e. the Warburg effect, anaplerotic TCA cycle flux and fatty acid and ketone body metabolism. The labeling at the C2 position has slightly increased proton coupling so HP [2-13C]pyruvate has a shorter T1 (~40-50 s at 3T) than [1-13C]pyruvate (34). Nevertheless, the T1 and percent polarization (~25%) are sufficient to enable clinical translation as detailed in section 8.4.
HP [2-13C]pyruvate has been used to monitor the response of glioblastoma tumors in vivo to DCA (76). Following injection of HP [2-13C]pyruvate into vehicle-treated rats bearing orthotopic C6 glioblastoma xenografts, the HP lactate/total carbon ratio was significantly higher in tumor relative to normal contralateral brain, the HP glutamate/total carbon was significantly lower in tumor versus normal brain and the HP lactate/glutamate ratio was higher in tumor relative to normal brain. These results are in line with the higher glycolysis and lower TCA cycle flux expected in tumors as compared to normal contralateral brain. Treatment with DCA induced a shift in this metabolic phenotype and resulted in a reduction in HP lactate/glutamate in tumor vs. normal contralateral brain. This reduction was comparable to the decrease in HP lactate/bicarbonate seen in studies with HP [1-13C]pyruvate (76).
IDHmut low-grade glioma differ from high-grade glioblastomas in the lack of a pronounced Warburg effect (10,79). However, the reprogramming of oxidative glucose metabolism via the TCA cycle follows the same pattern observed in glioblastomas and, indeed, most cancers (77,79). 13C-isotope labeling using [1-13C]glucose combined with thermally polarized 13C MRS showed reduced flux to glutamate in IDHmut cells relative to wild-type IDH cells, an effect associated with down-regulation of PDH activity. Importantly, HP [5-13C]glutamate production from HP [2-13C]pyruvate was reduced in IDHmut cells relative to wild-type IDH cells, providing a non-invasive method of monitoring IDHmut-induced reprogramming of PDH activity (77,79). Treatment with DCA upregulated PDH activity and inhibited tumorigenicity of IDHmut cells, identifying PDH as a therapeutic target for IDHmut gliomas. Concomitantly HP [5-13C]glutamate production from HP [2-13C]pyruvate was elevated, pointing to the use of HP [2-13C]pyruvate as a non-invasive imaging probe of response to DCA, and potentially emerging PDH-targeting therapeutics, in IDHmut low-grade gliomas (29,77,79).
Interrogating glutamine metabolism using HP [5-13C]glutamine or [1-13C]glutamate#
Glutamine is the second principal nutrient for mammalian cells and is a carbon and nitrogen donor for de novo biosynthesis of critical nitrogen-containing compounds including nucleotides and amino acids (2,3,5,7). Many tumor types display heightened glutaminolysis i.e. increased cellular uptake of glutamine and subsequent conversion to glutamate (see Figure 1). Glutamate in turn is in equilibrium with α-KG, which functions as an anaplerotic substrate in the TCA cycle. α-KG also plays a key role in epigenetic modulation of gene expression via its role as a cofactor for various dioxygenases including the Jmjc domain histone demethylases and the TET family of 5-methylcytosine hydroxylases (3,9,11).
It has long been recognized that tumors have a high demand for glutamine and several oncogenic signaling pathways including MYC, TP53 and the PI3K-Akt-mTOR pathway have been shown to modulate glutamine metabolism (Figure 1). Glutamine uptake and metabolism are generally elevated in tumors relative to corresponding normal tissue (2,3,5,7). 18F-labeled glutamine tracers that report on heightened tumor glutamine uptake have been developed and have shown promise in preclinical and early clinical studies, including in glioblastomas where the lack of background-to-tumor contrast limits the utility of FDG-PET imaging (80). HP 13C agents that track glutamine metabolism have the potential to provide complementary information on downstream glutamine metabolism.
Studies have examined the use of HP [5-13C]glutamine for probing glutamine metabolism in cancer (81-83). The major limitation of HP [5-13C]glutamine is spontaneous degradation of the HP preparation to [5-13C]glutamate, the expected product. Nevertheless, metabolism of HP [5-13C]glutamine to [5-13C]glutamate was higher in hepatoma cells relative to normal liver and reduced by chemotherapy with gemcitabine and etoposide (83). Another study also reported [5-13C]glutamate production from HP [5-13C]glutamine in liver tumor-bearing animals in vivo (81). [1-13C]glutamate has also been polarized and provides a potential alternate approach to interrogating glutamate metabolism to α-KG, which is central to TCA cycle flux and anaplerosis. Metabolism of HP [1-13C]glutamate to α-KG was observed in hepatoma cells and in vivo in a murine lymphoma model (84). Coadministration of sodium pyruvate increased HP [1-13C]α-KG levels, suggesting that intracellular pyruvate concentration is a limiting factor for HP [1-13C]glutamate metabolism (84). Further studies with HP [5-13C]glutamine and HP [1-13C]glutamate are needed to fully assess their utility for tumor imaging.
Interrogating oncometabolism using HP [1-13C]α-KG or [1-13C]glutamine#
The discovery of mutations in metabolic enzymes such as IDH underscored the critical role of metabolism in tumorigenesis and altered glioma classification. According to the latest WHO 2016 classification, tumors with IDHmut are now routinely classified as low-grade gliomas (85). IDHmut results in the production of a novel metabolite, 2-HG, which accumulates to concentrations of ~5-30 mM in low-grade gliomas (8). Several studies now indicate that 2-HG competitively inhibits the activity of a variety of α-KG-dependent dioxygenases, leading to epigenetic remodeling of the tumor genome (9-11,86). Due to its critical role, IDHmut is also a therapeutic target and IDHmut inhibitors are in clinical trials for gliomas (87-89). The ability to monitor α-KG metabolism to 2-HG via HP 13C metabolic imaging would, therefore, provide a non-invasive readout of tumor IDH status, thereby enabling tumor diagnosis and treatment response monitoring.
HP [1-13C]α-KG metabolism to [1-13C]2-HG has been visualized in genetically-engineered IDHmut glioma models (90). HP [1-13C]-α-KG has a T1 of ~45 s at 3T with 16% polarization. Following injection into cell lysates, metabolism to 2-HG was observed in lysates from IDHmut cells, but not wild-type IDH cells. Examination of rats bearing orthotopic tumor xenografts indicated that HP [1-13C]α-KG was detected in the tumor and blood of both IDHmut and wild-type IDH tumors while HP [1-13C]2-HG was only detected in IDHmut tumors, pointing to the potential of HP [1-13C]α-KG for detection of IDHmut status (90). In a separate study, HP [1-13C]α-KG metabolism to [1-13C]glutamate was shown to be reduced in IDHmut tumor relative to wild-type IDH tumors, an effect that was associated with reduced expression and activity of BCAT1, GOT1 and GDH, which are key enzymes involved in the interconversion of α-KG to glutamate (refer to Figure 8.1) (91).
HP [1-13C]glutamine metabolism to 2-HG has been examined in chondrosarcoma cells as well as tumors that express IDHmut (92). HP [1-13C]glutamine has a T1 of ~30 s with minimal degradation to glutamate. The chemical shift separation between [1-13C]glutamine (174.8 pm) and [1-13C]glutamate (175.5 ppm) makes it difficult to resolve these two species in vivo and, therefore, conversion to HP glutamate cannot be measured. Further studies are needed to determine the ability of HP [1-13C]-glutamine to monitor 2-HG production in IDHmut low-grade glioma models.
Imaging oncogenic reprogramming of metabolism using HP 13C MRS/I in the clinic#
In line with its widespread use in preclinical studies, HP [1-13C]pyruvate was the first HP 13C agent to transition to the clinic (33). Clinical trials with HP [1-13C]-pyruvate are currently underway in multiple tumor types in institutions across the globe (33). Given the poor utility of FDG-PET imaging for prostate and brain tumors, it is not surprising that the first clinical studies of HP [1-13C]pyruvate were conducted in these patient populations. The first-in-man study of HP [1-13C]pyruvate was carried out in prostate cancer patients at the University of California San Francisco (UCSF) (93). This landmark study established the safety and feasibility of using HP [1-13C]pyruvate for imaging tumor metabolism and thereby monitoring tumor burden. 31 patients with biopsy-proven prostate cancer were examined. Dose-limiting toxicities were absent at the maximum dose (~230 mM pyruvate) with the best SNR for HP [1-13C]pyruvate. Importantly, this study showed that the lactate to pyruvate ratio was elevated in biopsy-proven tumor regions relative to normal prostate and blood, thereby establishing the clinical utility of HP [1-13C]pyruvate in prostate cancer (93). A subsequent study examined the ability of HP [1-13C]pyruvate to monitor response to therapy in prostate cancer (94). HP [1-13C]pyruvate metabolism was examined in a patient with extensive high-grade prostate cancer before and 6 weeks after initiation of androgen ablation and chemotherapy. HP [1-13C]pyruvate to lactate conversion was elevated in tumor relative to adjacent normal prostate before treatment. Following 6 weeks of therapy, HP lactate production was almost completely abrogated while alterations in T2-weighted MRI were minimal. These results highlight the ability of HP [1-13C]pyruvate to provide an early readout of treatment response at a timepoint before changes can be detected using standard imaging criteria (94). Studies also suggest that HP lactate production from [1-13C]pyruvate in prostate cancer patients increases with Gleason grade in a manner that parallels increased MCT1 expression (95). A pilot clinical study also examined the utility of HP [1-13C]pyruvate in assessing prostate cancer metastases (96). Elevated HP [1-13C]pyruvate conversion to lactate was observed in metastatic tumors in the bone and liver, an effect associated with elevated expression of LDHA and reduced expression of the PDH isoform PDHA1. Importantly, reduced pyruvate to lactate conversion was noted 2 months after initiation of taxane plus platinum chemotherapy (96).
The first study in brain tumors was carried out in 8 patients at UCSF with a confirmed diagnosis of glioma (4 high-grade primary glioblastoma, 3 low-grade oligodendroglioma and 1 low-grade astrocytoma) (97). As with prostate cancer patients, dose-limiting toxicities were not observed in any of the patients. Tumor regions showed the presence of lactate while both lactate and bicarbonate were observed in normal contralateral brain, pointing to sufficient delivery of HP [1-13C]pyruvate across the BBB. However, unlike prostate cancer, the lactate/pyruvate ratio in tumor voxels were similar to, or in some cases lower than, surrounding normal contralateral brain. Nevertheless, the bicarbonate to lactate ratio was able to differentiate tumor from normal brain (97). These results were confirmed by another study that also included patients with metastatic brain tumors (98). Lactate production from HP [1-13C]pyruvate was similar between tumor and contralateral brain in primary brain tumor patients. The observation of high pyruvate to lactate conversion in normal human brain in these early clinical studies is in contrast to the body of data available from preclinical studies (refer to section 8.3.1) that showed very little lactate production in the normal rodent brain. High background lactate production in the normal human brain has also been confirmed in healthy human volunteers (99,100). These clinical results highlight the need to build a better understanding of glycolytic metabolism and its regulation in the human brain. In the context of brain tumors, it is important to note that a recent study showed that patient-derived glioblastoma tumors display pyruvate to lactate conversion that is comparable to normal brain (56). Further studies are needed to fully determine the utility of the HP bicarbonate/lactate ratio in monitoring tumor burden and response to therapy in glioma patients.
A recent study assessed the potential utility of HP [1-13C]pyruvate in imaging metabolism in breast cancer patients (101). HP lactate production from pyruvate was observed in patients with triple negative breast cancer or higher-grade (grade 3) tumors. In contrast, there was no detectable pyruvate to lactate conversion in patients with lower-grade (grade 2) tumors. This study pointed to the presence of occurrence of considerable intertumoral and intratumoral heterogeneity in the observed HP [1-13C]pyruvate and [1-13C]lactate signals. Mechanistically, conversion to lactate was correlated with MCT1 expression and hypoxia (101).
A pilot clinical study in 2 patients recently tested the utility of HP [1-13C]pyruvate in pancreatic cancer. In both patients, production of HP [1-13C]alanine and HP [1-13C]lactate was observed in the tumor and served to differentiate tumor from surrounding normal tissues (102). Interestingly, unlike preclinical studies in genetically-engineered models of pancreatic ductal adenocarcinoma which indicated a decrease in the alanine/lactate signal ratio with disease progression (38), both alanine and lactate were higher in this clinical study relative to normal pancreas (102). While further studies are needed to fully determine the utility of HP pyruvate in imaging pancreatic cancer patients, these results point to the promising potential of HP [1-13C]pyruvate in discriminating pancreatic tumor from normal pancreas.
Assessment of HP [1-13C]pyruvate metabolism in a pilot study in RCC patients showed heterogenous lactate production across the tumor region (103). Importantly, HP lactate production was correlated with ex vivo liquid chromatography-mass spectrometric analysis of lactate levels, which also showed heterogeneity across the tumor. Further studies are warranted to assess whether this metabolic heterogeneity reflects underlying genomic heterogeneity in RCC.
Clinical translation of HP [2-13C]pyruvate is currently underway (104,105). A study in 4 healthy human volunteers showed dynamic production of [2-13C]lactate and [5-13C]glutamate from HP [2-13C]pyruvate (105). Based on the preclinical studies in IDHmut low-grade glioma models (see section 8.3.2) (77,79), clinical studies on IDHmut low-grade glioma patients have been initiated (104). Initial results in 2 patients indicate that tumor lesions display a reduced [5-13C] glutamate/[2-13C]pyruvate ratio and an elevated [5-13C]glutamate/[5-13C]glutamine ratio relative to normal brain, pointing to the promising potential of HP [2-13C]pyruvate in glioma patients (104).
Conclusions#
Oncogenic signaling pathways lead to metabolic reprogramming, which is now recognized as a fundamental hallmark of cancer. Defining alterations in tumor metabolism provides a framework for the identification of non-invasive metabolic imaging biomarkers and therapeutic targets. HP 13C MRS/I is a non-invasive metabolic imaging modality that offers the ability to visualize metabolic fluxes in real time. As summarized in Table 1, several HP 13C agents have been identified that allow interrogation of various aspects of oncogenic metabolic reprogramming in preclinical studies. Further studies are needed to integrate these HP 13C agents into clinical practice and establish their utility for tumor diagnosis, prognosis, patient stratification and treatment response monitoring.
Acknowledgements
This work was supported by the following funding sources: NIH R01CA239288 and Department of Defense W81XWH201055315.
References#
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011;144(5):646-674.
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell metabolism 2008;7(1):11-20.
Pavlova NN, Thompson CB. The Emerging Hallmarks of Cancer Metabolism. Cell metabolism 2016;23(1):27-47.
Hirschey MD, DeBerardinis RJ, Diehl AM, Drew JE, Frezza C, Green MF, Jones LW, Ko YH, Le A, Lea MA, Locasale JW, Longo VD, Lyssiotis CA, McDonnell E, Mehrmohamadi M, Michelotti G, Muralidhar V, Murphy MP, Pedersen PL, Poore B, Raffaghello L, Rathmell JC, Sivanand S, Heiden MG, Wellen KE. Dysregulated metabolism contributes to oncogenesis. Seminars in cancer biology 2015.
Hensley CT, Wasti AT, DeBerardinis RJ. Glutamine and cancer: cell biology, physiology, and clinical opportunities. The Journal of clinical investigation 2013;123(9):3678-3684.
Mullen AR, DeBerardinis RJ. Genetically-defined metabolic reprogramming in cancer. Trends in endocrinology and metabolism: TEM 2012;23(11):552-559.
DeBerardinis RJ, Chandel NS. Fundamentals of cancer metabolism. Sci Adv 2016;2(5):e1600200.
Dang L, White DW, Gross S, Bennett BD, Bittinger MA, Driggers EM, Fantin VR, Jang HG, Jin S, Keenan MC, Marks KM, Prins RM, Ward PS, Yen KE, Liau LM, Rabinowitz JD, Cantley LC, Thompson CB, Vander Heiden MG, Su SM. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 2009;462(7274):739-744.
Turcan S, Rohle D, Goenka A, Walsh LA, Fang F, Yilmaz E, Campos C, Fabius AW, Lu C, Ward PS, Thompson CB, Kaufman A, Guryanova O, Levine R, Heguy A, Viale A, Morris LG, Huse JT, Mellinghoff IK, Chan TA. IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature 2012;483(7390):479-483.
Viswanath P, Chaumeil MM, Ronen SM. Molecular Imaging of Metabolic Reprograming in Mutant IDH Cells. Frontiers in oncology 2016;6:60.
Lu C, Ward PS, Kapoor GS, Rohle D, Turcan S, Abdel-Wahab O, Edwards CR, Khanin R, Figueroa ME, Melnick A, Wellen KE, O’Rourke DM, Berger SL, Chan TA, Levine RL, Mellinghoff IK, Thompson CB. IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature 2012;483(7390):474-478.
O’Connor JP, Aboagye EO, Adams JE, Aerts HJ, Barrington SF, Beer AJ, Boellaard R, Bohndiek SE, Brady M, Brown G, Buckley DL, Chenevert TL, Clarke LP, Collette S, Cook GJ, deSouza NM, Dickson JC, Dive C, Evelhoch JL, Faivre-Finn C, Gallagher FA, Gilbert FJ, Gillies RJ, Goh V, Griffiths JR, Groves AM, Halligan S, Harris AL, Hawkes DJ, Hoekstra OS, Huang EP, Hutton BF, Jackson EF, Jayson GC, Jones A, Koh DM, Lacombe D, Lambin P, Lassau N, Leach MO, Lee TY, Leen EL, Lewis JS, Liu Y, Lythgoe MF, Manoharan P, Maxwell RJ, Miles KA, Morgan B, Morris S, Ng T, Padhani AR, Parker GJ, Partridge M, Pathak AP, Peet AC, Punwani S, Reynolds AR, Robinson SP, Shankar LK, Sharma RA, Soloviev D, Stroobants S, Sullivan DC, Taylor SA, Tofts PS, Tozer GM, van Herk M, Walker-Samuel S, Wason J, Williams KJ, Workman P, Yankeelov TE, Brindle KM, McShane LM, Jackson A, Waterton JC. Imaging biomarker roadmap for cancer studies. Nat Rev Clin Oncol 2017;14(3):169-186.
Ellingson BM, Bendszus M, Boxerman J, Barboriak D, Erickson BJ, Smits M, Nelson SJ, Gerstner E, Alexander B, Goldmacher G, Wick W, Vogelbaum M, Weller M, Galanis E, Kalpathy-Cramer J, Shankar L, Jacobs P, Pope WB, Yang D, Chung C, Knopp MV, Cha S, van den Bent MJ, Chang S, Yung WK, Cloughesy TF, Wen PY, Gilbert MR. Consensus recommendations for a standardized Brain Tumor Imaging Protocol in clinical trials. Neuro-oncology 2015;17(9):1188-1198.
Weissleder R, Pittet MJ. Imaging in the era of molecular oncology. Nature 2008;452:580.
Schwartz LH, Litière S, de Vries E, Ford R, Gwyther S, Mandrekar S, Shankar L, Bogaerts J, Chen A, Dancey J, Hayes W, Hodi FS, Hoekstra OS, Huang EP, Lin N, Liu Y, Therasse P, Wolchok JD, Seymour L. RECIST 1.1-Update and clarification: From the RECIST committee. European journal of cancer (Oxford, England : 1990) 2016;62:132-137.
Brindle K. New approaches for imaging tumour responses to treatment. Nature reviews Cancer 2008;8(2):94-107.
Shin SH, Bode AM, Dong Z. Precision medicine: the foundation of future cancer therapeutics. npj Precision Oncology 2017;1(1):12.
Deng X, Nakamura Y. Cancer Precision Medicine: From Cancer Screening to Drug Selection and Personalized Immunotherapy. Trends Pharmacol Sci 2017;38(1):15-24.
Oellerich M, Schütz E, Beck J, Walson PD. Circulating Cell-Free DNA-Diagnostic and Prognostic Applications in Personalized Cancer Therapy. Ther Drug Monit 2019;41(2):115-120.
Miller AM, Shah RH, Pentsova EI, Pourmaleki M, Briggs S, Distefano N, Zheng Y, Skakodub A, Mehta SA, Campos C, Hsieh WY, Selcuklu SD, Ling L, Meng F, Jing X, Samoila A, Bale TA, Tsui DWY, Grommes C, Viale A, Souweidane MM, Tabar V, Brennan CW, Reiner AS, Rosenblum M, Panageas KS, DeAngelis LM, Young RJ, Berger MF, Mellinghoff IK. Tracking tumour evolution in glioma through liquid biopsies of cerebrospinal fluid. Nature 2019;565(7741):654-658.
Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 2009;324(5930):1029-1033.
Zhu A, Lee D, Shim H. Metabolic PET Imaging in Cancer Detection and Therapy Response. Semin Oncol 2011;38(1):55-69.
Fletcher JW, Djulbegovic B, Soares HP, Siegel BA, Lowe VJ, Lyman GH, Coleman RE, Wahl R, Paschold JC, Avril N, Einhorn LH, Suh WW, Samson D, Delbeke D, Gorman M, Shields AF. Recommendations on the use of 18F-FDG PET in oncology. Journal of nuclear medicine : official publication, Society of Nuclear Medicine 2008;49(3):480-508.
Gillies RJ, Morse DL. In vivo magnetic resonance spectroscopy in cancer. Annual review of biomedical engineering 2005;7:287-326.
Glunde K, Bhujwalla ZM. Metabolic Tumor Imaging Using Magnetic Resonance Spectroscopy. Seminars in Oncology 2011;38(1):26-41.
Rodrigues TB, Cerdán S. 13C MRS: An outstanding tool for metabolic studies. Concepts in Magnetic Resonance Part A 2005;27A(1):1-16.
Rothman DL, De Feyter HM, de Graaf RA, Mason GF, Behar KL. (13)C MRS studies of neuroenergetics and neurotransmitter cycling in humans. NMR in biomedicine 2011;24(8):943-957.
Lumata L, Yang C, Ragavan M, Carpenter N, DeBerardinis RJ, Merritt ME. Hyperpolarized (13)C Magnetic Resonance and Its Use in Metabolic Assessment of Cultured Cells and Perfused Organs. Methods Enzymol 2015;561:73-106.
Viswanath P, Li Y, Ronen SM. C-13 Hyperpolarized MR Spectroscopy for Metabolic Imaging of Brain Tumors. In: Pope WB, editor. Glioma Imaging: Physiologic, Metabolic, and Molecular Approaches. Cham: Springer International Publishing; 2020. p 191-209.
Lunt SY, Vander Heiden MG. Aerobic glycolysis: meeting the metabolic requirements of cell proliferation. Annual review of cell and developmental biology 2011;27:441-464.
de la Cruz-López KG, Castro-Muñoz LJ, Reyes-Hernández DO, García-Carrancá A, Manzo-Merino J. Lactate in the Regulation of Tumor Microenvironment and Therapeutic Approaches. Frontiers in oncology 2019;9:1143.
Semenza GL. HIF-1 mediates metabolic responses to intratumoral hypoxia and oncogenic mutations. The Journal of clinical investigation 2013;123(9):3664-3671.
Kurhanewicz J, Vigneron DB, Ardenkjaer-Larsen JH, Bankson JA, Brindle K, Cunningham CH, Gallagher FA, Keshari KR, Kjaer A, Laustsen C, Mankoff DA, Merritt ME, Nelson SJ, Pauly JM, Lee P, Ronen S, Tyler DJ, Rajan SS, Spielman DM, Wald L, Zhang X, Malloy CR, Rizi R. Hyperpolarized (13)C MRI: Path to Clinical Translation in Oncology. Neoplasia (New York, NY) 2019;21(1):1-16.
Wang ZJ, Ohliger MA, Larson PEZ, Gordon JW, Bok RA, Slater J, Villanueva-Meyer JE, Hess CP, Kurhanewicz J, Vigneron DB. Hyperpolarized (13)C MRI: State of the Art and Future Directions. Radiology 2019;291(2):273-284.
Halestrap AP. The monocarboxylate transporter family–Structure and functional characterization. IUBMB life 2012;64(1):1-9.
Smith B, Schafer XL, Ambeskovic A, Spencer CM, Land H, Munger J. Addiction to Coupling of the Warburg Effect with Glutamine Catabolism in Cancer Cells. Cell reports 2016;17(3):821-836.
Harris T, Eliyahu G, Frydman L, Degani H. Kinetics of hyperpolarized 13C1-pyruvate transport and metabolism in living human breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America 2009;106(43):18131-18136.
Serrao EM, Kettunen MI, Rodrigues TB, Dzien P, Wright AJ, Gopinathan A, Gallagher FA, Lewis DY, Frese KK, Almeida J, Howat WJ, Tuveson DA, Brindle KM. MRI with hyperpolarised [1-13C]pyruvate detects advanced pancreatic preneoplasia prior to invasive disease in a mouse model. Gut 2016;65(3):465-475.
Witney TH, Kettunen MI, Brindle KM. Kinetic modeling of hyperpolarized 13C label exchange between pyruvate and lactate in tumor cells. The Journal of biological chemistry 2011;286(28):24572-24580.
Day SE, Kettunen MI, Gallagher FA, Hu DE, Lerche M, Wolber J, Golman K, Ardenkjaer-Larsen JH, Brindle KM. Detecting tumor response to treatment using hyperpolarized 13C magnetic resonance imaging and spectroscopy. Nat Med 2007;13(11):1382-1387.
Welch HG, Black WC. Overdiagnosis in Cancer. JNCI: Journal of the National Cancer Institute 2010;102(9):605-613.
Cooperberg MR. Long-term active surveillance for prostate cancer: answers and questions. J Clin Oncol 2015;33(3):238-240.
Albers MJ, Bok R, Chen AP, Cunningham CH, Zierhut ML, Zhang VY, Kohler SJ, Tropp J, Hurd RE, Yen YF, Nelson SJ, Vigneron DB, Kurhanewicz J. Hyperpolarized 13C lactate, pyruvate, and alanine: noninvasive biomarkers for prostate cancer detection and grading. Cancer research 2008;68(20):8607-8615.
Keshari KR, Sriram R, Van Criekinge M, Wilson DM, Wang ZJ, Vigneron DB, Peehl DM, Kurhanewicz J. Metabolic reprogramming and validation of hyperpolarized 13C lactate as a prostate cancer biomarker using a human prostate tissue slice culture bioreactor. The Prostate 2013;73(11):1171-1181.
Sriram R, Gordon J, Baligand C, Ahamed F, Delos Santos J, Qin H, Bok RA, Vigneron DB, Kurhanewicz J, Larson PEZ, Wang ZJ. Non-Invasive Assessment of Lactate Production and Compartmentalization in Renal Cell Carcinomas Using Hyperpolarized (13)C Pyruvate MRI. Cancers (Basel) 2018;10(9).
Sriram R, Van Criekinge M, Hansen A, Wang ZJ, Vigneron DB, Wilson DM, Keshari KR, Kurhanewicz J. Real-time measurement of hyperpolarized lactate production and efflux as a biomarker of tumor aggressiveness in an MR compatible 3D cell culture bioreactor. NMR in biomedicine 2015;28(9):1141-1149.
Sriram R, Van Criekinge M, DeLos Santos J, Keshari KR, Wilson DM, Peehl D, Kurhanewicz J, Wang ZJ. Non-invasive differentiation of benign renal tumors from clear cell renal cell carcinomas using clinically translatable hyperpolarized (13)C pyruvate magnetic resonance. Tomography 2016;2(1):35-42.
Hollingsworth JM, Miller DC, Daignault S, Hollenbeck BK. Rising incidence of small renal masses: a need to reassess treatment effect. Journal of the National Cancer Institute 2006;98(18):1331-1334.
Rawla P, Sunkara T, Gaduputi V. Epidemiology of Pancreatic Cancer: Global Trends, Etiology and Risk Factors. World J Oncol 2019;10(1):10-27.
Canto MI, Harinck F, Hruban RH, Offerhaus GJ, Poley JW, Kamel I, Nio Y, Schulick RS, Bassi C, Kluijt I, Levy MJ, Chak A, Fockens P, Goggins M, Bruno M. International Cancer of the Pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 2013;62(3):339-347.
Hu S, Balakrishnan A, Bok RA, Anderton B, Larson PE, Nelson SJ, Kurhanewicz J, Vigneron DB, Goga A. 13C-pyruvate imaging reveals alterations in glycolysis that precede c-Myc-induced tumor formation and regression. Cell metabolism 2011;14(1):131-142.
Taouli B, Losada M, Holland A, Krinsky G. Magnetic resonance imaging of hepatocellular carcinoma. Gastroenterology 2004;127(5 Suppl 1):S144-152.
Shin PJ, Zhu Z, Camarda R, Bok RA, Zhou AY, Kurhanewicz J, Goga A, Vigneron DB. Cancer recurrence monitoring using hyperpolarized [1-(13)C]pyruvate metabolic imaging in murine breast cancer model. Magnetic resonance imaging 2017;43:105-109.
Park I, Hu S, Bok R, Ozawa T, Ito M, Mukherjee J, Phillips JJ, James CD, Pieper RO, Ronen SM, Vigneron DB, Nelson SJ. Evaluation of heterogeneous metabolic profile in an orthotopic human glioblastoma xenograft model using compressed sensing hyperpolarized 3D 13C magnetic resonance spectroscopic imaging. Magn Reson Med 2013;70(1):33-39.
Park I, Larson PE, Zierhut ML, Hu S, Bok R, Ozawa T, Kurhanewicz J, Vigneron DB, Vandenberg SR, James CD, Nelson SJ. Hyperpolarized 13C magnetic resonance metabolic imaging: application to brain tumors. Neuro-oncology 2010;12(2):133-144.
Mair R, Wright AJ, Ros S, Hu DE, Booth T, Kreis F, Rao J, Watts C, Brindle KM. Metabolic Imaging Detects Low Levels of Glycolytic Activity That Vary with Levels of c-Myc Expression in Patient-Derived Xenograft Models of Glioblastoma. Cancer research 2018;78(18):5408-5418.
Park JM, Recht LD, Josan S, Merchant M, Jang T, Yen YF, Hurd RE, Spielman DM, Mayer D. Metabolic response of glioma to dichloroacetate measured in vivo by hyperpolarized (13)C magnetic resonance spectroscopic imaging. Neuro-oncology 2013;15(4):433-441.
Viswanath P, Najac C, Izquierdo-Garcia JL, Pankov A, Hong C, Eriksson P, Costello JF, Pieper RO, Ronen SM. Mutant IDH1 expression is associated with down-regulation of monocarboxylate transporters. Oncotarget 2016;7(23):34942-34955.
Chesnelong C, Chaumeil MM, Blough MD, Al-Najjar M, Stechishin OD, Chan JA, Pieper RO, Ronen SM, Weiss S, Luchman HA, Cairncross JG. Lactate dehydrogenase A silencing in IDH mutant gliomas. Neuro-oncology 2014;16(5):686-695.
Chaumeil MM, Radoul M, Najac C, Eriksson P, Viswanath P, Blough MD, Chesnelong C, Luchman HA, Cairncross JG, Ronen SM. Hyperpolarized (13)C MR imaging detects no lactate production in mutant IDH1 gliomas: Implications for diagnosis and response monitoring. NeuroImage Clinical 2016;12:180-189.
Victor RR, Malta TM, Seki T, Lita A, Dowdy T, Celiku O, Cavazos-Saldana A, Li A, Liu Y, Han S, Zhang W, Song H, Davis D, Lee S, Trepel JB, Sabedot TS, Munasinghe J, Yang C, Herold-Mende C, Gilbert MR, Krishna Cherukuri M, Noushmehr H, Larion M. Metabolic Reprogramming Associated with Aggressiveness Occurs in the G-CIMP-High Molecular Subtypes of IDH1mut Lower Grade Gliomas. Neuro-oncology 2019.
Day SE, Kettunen MI, Cherukuri MK, Mitchell JB, Lizak MJ, Morris HD, Matsumoto S, Koretsky AP, Brindle KM. Detecting response of rat C6 glioma tumors to radiotherapy using hyperpolarized [1- 13C]pyruvate and 13C magnetic resonance spectroscopic imaging. Magn Reson Med 2011;65(2):557-563.
Park I, Mukherjee J, Ito M, Chaumeil MM, Jalbert LE, Gaensler K, Ronen SM, Nelson SJ, Pieper RO. Changes in pyruvate metabolism detected by magnetic resonance imaging are linked to DNA damage and serve as a sensor of temozolomide response in glioblastoma cells. Cancer research 2014;74(23):7115-7124.
Rodon J, Dienstmann R, Serra V, Tabernero J. Development of PI3K inhibitors: lessons learned from early clinical trials. Nat Rev Clin Oncol 2013;10(3):143-153.
Courtney KD, Corcoran RB, Engelman JA. The PI3K pathway as drug target in human cancer. J Clin Oncol 2010;28(6):1075-1083.
Venkatesh HS, Chaumeil MM, Ward CS, Haas-Kogan DA, James CD, Ronen SM. Reduced phosphocholine and hyperpolarized lactate provide magnetic resonance biomarkers of PI3K/Akt/mTOR inhibition in glioblastoma. Neuro-oncology 2012;14(3):315-325.
Ward CS, Venkatesh HS, Chaumeil MM, Brandes AH, Vancriekinge M, Dafni H, Sukumar S, Nelson SJ, Vigneron DB, Kurhanewicz J, James CD, Haas-Kogan DA, Ronen SM. Noninvasive detection of target modulation following phosphatidylinositol 3-kinase inhibition using hyperpolarized 13C magnetic resonance spectroscopy. Cancer research 2010;70(4):1296-1305.
Radoul M, Chaumeil MM, Eriksson P, Wang AS, Phillips JJ, Ronen SM. MR Studies of Glioblastoma Models Treated with Dual PI3K/mTOR Inhibitor and Temozolomide:Metabolic Changes Are Associated with Enhanced Survival. Molecular cancer therapeutics 2016;15(5):1113-1122.
Chaumeil MM, Ozawa T, Park I, Scott K, James CD, Nelson SJ, Ronen SM. Hyperpolarized 13C MR spectroscopic imaging can be used to monitor Everolimus treatment in vivo in an orthotopic rodent model of glioblastoma. Neuroimage 2012;59(1):193-201.
Park JM, Spielman DM, Josan S, Jang T, Merchant M, Hurd RE, Mayer D, Recht LD. Hyperpolarized (13)C-lactate to (13)C-bicarbonate ratio as a biomarker for monitoring the acute response of anti-vascular endothelial growth factor (anti-VEGF) treatment. NMR in biomedicine 2016;29(5):650-659.
Michelakis ED, Sutendra G, Dromparis P, Webster L, Haromy A, Niven E, Maguire C, Gammer T-L, Mackey JR, Fulton D, Abdulkarim B, McMurtry MS, Petruk KC. Metabolic Modulation of Glioblastoma with Dichloroacetate. Science translational medicine 2010;2(31):31ra34.
Qin H, Zhang V, Bok RA, Santos RD, Cunha JA, Hsu IC, Santos Bs JD, Lee JE, Sukumar S, Larson PEZ, Vigneron DB, Wilson DM, Sriram R, Kurhanewicz J. Simultaneous Metabolic and Perfusion Imaging Using Hyperpolarized (13)C MRI Can Evaluate Early and Dose-Dependent Response to Radiation Therapy in a Prostate Cancer Mouse Model. Int J Radiat Oncol Biol Phys 2020.
Saito K, Matsumoto S, Takakusagi Y, Matsuo M, Morris HD, Lizak MJ, Munasinghe JP, Devasahayam N, Subramanian S, Mitchell JB, Krishna MC. 13C-MR Spectroscopic Imaging with Hyperpolarized [1-13C]pyruvate Detects Early Response to Radiotherapy in SCC Tumors and HT-29 Tumors. Clinical cancer research : an official journal of the American Association for Cancer Research 2015;21(22):5073-5081.
Hesketh RL, Wang J, Wright AJ, Lewis DY, Denton AE, Grenfell R, Miller JL, Bielik R, Gehrung M, Fala M, Ros S, Xie B, Hu DE, Brindle KM. Magnetic Resonance Imaging Is More Sensitive Than PET for Detecting Treatment-Induced Cell Death-Dependent Changes in Glycolysis. Cancer research 2019;79(14):3557-3569.
Park JM, Josan S, Grafendorfer T, Yen Y-F, Hurd RE, Spielman DM, Mayer D. Measuring mitochondrial metabolism in rat brain in vivo using MR Spectroscopy of hyperpolarized [2-13C]pyruvate. NMR in biomedicine 2013;26(10):1197-1203.
Park JM, Josan S, Jang T, Merchant M, Watkins R, Hurd RE, Recht LD, Mayer D, Spielman DM. Volumetric spiral chemical shift imaging of hyperpolarized [2-(13) c]pyruvate in a rat c6 glioma model. Magn Reson Med 2016;75(3):973-984.
Izquierdo-Garcia JL, Viswanath P, Eriksson P, Cai L, Radoul M, Chaumeil MM, Blough M, Luchman HA, Weiss S, Cairncross JG, Philips JJ, Pieper RO, Ronen SM. IDH1 mutation induces reprogramming of pyruvate metabolism. Cancer research 2015.
Roche TE, Hiromasa Y. Pyruvate dehydrogenase kinase regulatory mechanisms and inhibition in treating diabetes, heart ischemia, and cancer. Cellular and molecular life sciences : CMLS 2007;64(7-8):830-849.
Viswanath P, Ronen SM. Metabolic reprogramming of pyruvate dehydrogenase is essential for the proliferation of glioma cells expressing mutant IDH1. Molecular & cellular oncology 2016;3(2):e1077922.
Venneti S, Dunphy MP, Zhang H, Pitter KL, Zanzonico P, Campos C, Carlin SD, La Rocca G, Lyashchenko S, Ploessl K, Rohle D, Omuro AM, Cross JR, Brennan CW, Weber WA, Holland EC, Mellinghoff IK, Kung HF, Lewis JS, Thompson CB. Glutamine-based PET imaging facilitates enhanced metabolic evaluation of gliomas in vivo. Science translational medicine 2015;7(274):274ra217.
Cabella C, Karlsson M, Canape C, Catanzaro G, Colombo Serra S, Miragoli L, Poggi L, Uggeri F, Venturi L, Jensen PR, Lerche MH, Tedoldi F. In vivo and in vitro liver cancer metabolism observed with hyperpolarized [5-(13)C]glutamine. Journal of magnetic resonance (San Diego, Calif : 1997) 2013;232:45-52.
Canape C, Catanzaro G, Terreno E, Karlsson M, Lerche MH, Jensen PR. Probing treatment response of glutaminolytic prostate cancer cells to natural drugs with hyperpolarized [5-(13) C]glutamine. Magn Reson Med 2015;73(6):2296-2305.
Gallagher FA, Kettunen MI, Day SE, Lerche M, Brindle KM. 13C MR spectroscopy measurements of glutaminase activity in human hepatocellular carcinoma cells using hyperpolarized 13C-labeled glutamine. Magn Reson Med 2008;60(2):253-257.
Gallagher FA, Kettunen MI, Day SE, Hu DE, Karlsson M, Gisselsson A, Lerche MH, Brindle KM. Detection of tumor glutamate metabolism in vivo using (13)C magnetic resonance spectroscopy and hyperpolarized [1-(13)C]glutamate. Magn Reson Med 2011;66(1):18-23.
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 2016;131(6):803-820.
Flavahan WA, Drier Y, Liau BB, Gillespie SM, Venteicher AS, Stemmer-Rachamimov AO, Suva ML, Bernstein BE. Insulator dysfunction and oncogene activation in IDH mutant gliomas. Nature 2016;529(7584):110-114.
Dang L, Yen K, Attar EC. IDH mutations in cancer and progress toward development of targeted therapeutics. Annals of oncology : official journal of the European Society for Medical Oncology 2016;27(4):599-608.
Pusch S, Krausert S, Fischer V, Balss J, Ott M, Schrimpf D, Capper D, Sahm F, Eisel J, Beck AC, Jugold M, Eichwald V, Kaulfuss S, Panknin O, Rehwinkel H, Zimmermann K, Hillig RC, Guenther J, Toschi L, Neuhaus R, Haegebart A, Hess-Stumpp H, Bauser M, Wick W, Unterberg A, Herold-Mende C, Platten M, von Deimling A. Pan-mutant IDH1 inhibitor BAY 1436032 for effective treatment of IDH1 mutant astrocytoma in vivo. Acta Neuropathol 2017;133(4):629-644.
Rohle D, Popovici-Muller J, Palaskas N, Turcan S, Grommes C, Campos C, Tsoi J, Clark O, Oldrini B, Komisopoulou E, Kunii K, Pedraza A, Schalm S, Silverman L, Miller A, Wang F, Yang H, Chen Y, Kernytsky A, Rosenblum MK, Liu W, Biller SA, Su SM, Brennan CW, Chan TA, Graeber TG, Yen KE, Mellinghoff IK. An inhibitor of mutant IDH1 delays growth and promotes differentiation of glioma cells. Science 2013;340(6132):626-630.
Chaumeil MM, Larson PE, Yoshihara HA, Danforth OM, Vigneron DB, Nelson SJ, Pieper RO, Phillips JJ, Ronen SM. Non-invasive in vivo assessment of IDH1 mutational status in glioma. Nature communications 2013;4:2429.
Chaumeil MM, Larson PE, Woods SM, Cai L, Eriksson P, Robinson AE, Lupo JM, Vigneron DB, Nelson SJ, Pieper RO, Phillips JJ, Ronen SM. Hyperpolarized [1-13C] glutamate: a metabolic imaging biomarker of IDH1 mutational status in glioma. Cancer research 2014;74(16):4247-4257.
Salamanca-Cardona L, Shah H, Poot AJ, Correa FM, Di Gialleonardo V, Lui H, Miloushev VZ, Granlund KL, Tee SS, Cross JR, Thompson CB, Keshari KR. In Vivo Imaging of Glutamine Metabolism to the Oncometabolite 2-Hydroxyglutarate in IDH1/2 Mutant Tumors. Cell metabolism 2017;26(6):830-841 e833.
Nelson SJ, Kurhanewicz J, Vigneron DB, Larson PE, Harzstark AL, Ferrone M, van Criekinge M, Chang JW, Bok R, Park I, Reed G, Carvajal L, Small EJ, Munster P, Weinberg VK, Ardenkjaer-Larsen JH, Chen AP, Hurd RE, Odegardstuen LI, Robb FJ, Tropp J, Murray JA. Metabolic imaging of patients with prostate cancer using hyperpolarized [1-(1)(3)C]pyruvate. Science translational medicine 2013;5(198):198ra108.
Aggarwal R, Vigneron DB, Kurhanewicz J. Hyperpolarized 1-[(13)C]-Pyruvate Magnetic Resonance Imaging Detects an Early Metabolic Response to Androgen Ablation Therapy in Prostate Cancer. Eur Urol 2017;72(6):1028-1029.
Granlund KL, Tee SS, Vargas HA, Lyashchenko SK, Reznik E, Fine S, Laudone V, Eastham JA, Touijer KA, Reuter VE, Gonen M, Sosa RE, Nicholson D, Guo YW, Chen AP, Tropp J, Robb F, Hricak H, Keshari KR. Hyperpolarized MRI of Human Prostate Cancer Reveals Increased Lactate with Tumor Grade Driven by Monocarboxylate Transporter 1. Cell metabolism 2020;31(1):105-114.e103.
Chen HY, Aggarwal R, Bok RA, Ohliger MA, Zhu Z, Lee P, Gordon JW, van Criekinge M, Carvajal L, Slater JB, Larson PEZ, Small EJ, Kurhanewicz J, Vigneron DB. Hyperpolarized (13)C-pyruvate MRI detects real-time metabolic flux in prostate cancer metastases to bone and liver: a clinical feasibility study. Prostate Cancer Prostatic Dis 2020;23(2):269-276.
Park I, Larson PEZ, Gordon JW, Carvajal L, Chen HY, Bok R, Van Criekinge M, Ferrone M, Slater JB, Xu D, Kurhanewicz J, Vigneron DB, Chang S, Nelson SJ. Development of methods and feasibility of using hyperpolarized carbon-13 imaging data for evaluating brain metabolism in patient studies. Magn Reson Med 2018.
Miloushev VZ, Granlund KL, Boltyanskiy R, Lyashchenko SK, DeAngelis LM, Mellinghoff IK, Brennan CW, Tabar V, Yang TJ, Holodny AI, Sosa RE, Guo YW, Chen AP, Tropp J, Robb F, Keshari KR. Metabolic Imaging of the Human Brain with Hyperpolarized 13C Pyruvate Demonstrates 13C Lactate Production in Brain Tumor Patients. Cancer research 2018.
Grist JT, McLean MA, Riemer F, Schulte RF, Deen SS, Zaccagna F, Woitek R, Daniels CJ, Kaggie JD, Matys T, Patterson I, Slough R, Gill AB, Chhabra A, Eichenberger R, Laurent MC, Comment A, Gillard JH, Coles AJ, Tyler DJ, Wilkinson I, Basu B, Lomas DJ, Graves MJ, Brindle KM, Gallagher FA. Quantifying normal human brain metabolism using hyperpolarized [1-(13)C]pyruvate and magnetic resonance imaging. Neuroimage 2019;189:171-179.
Lee CY, Soliman H, Geraghty BJ, Chen AP, Connelly KA, Endre R, Perks WJ, Heyn C, Black SE, Cunningham CH. Lactate topography of the human brain using hyperpolarized (13)C-MRI. Neuroimage 2020;204:116202.
Gallagher FA, Woitek R, McLean MA, Gill AB, Manzano Garcia R, Provenzano E, Riemer F, Kaggie J, Chhabra A, Ursprung S, Grist JT, Daniels CJ, Zaccagna F, Laurent MC, Locke M, Hilborne S, Frary A, Torheim T, Boursnell C, Schiller A, Patterson I, Slough R, Carmo B, Kane J, Biggs H, Harrison E, Deen SS, Patterson A, Lanz T, Kingsbury Z, Ross M, Basu B, Baird R, Lomas DJ, Sala E, Wason J, Rueda OM, Chin SF, Wilkinson IB, Graves MJ, Abraham JE, Gilbert FJ, Caldas C, Brindle KM. Imaging breast cancer using hyperpolarized carbon-13 MRI. Proceedings of the National Academy of Sciences of the United States of America 2020;117(4):2092-2098.
Stødkilde-Jørgensen H, Laustsen C, Hansen ESS, Schulte R, Ardenkjaer-Larsen JH, Comment A, Frøkiaer J, Ringgaard S, Bertelsen LB, Ladekarl M, Weber B. Pilot Study Experiences With Hyperpolarized [1-(13) C]pyruvate MRI in Pancreatic Cancer Patients. Journal of magnetic resonance imaging : JMRI 2020;51(3):961-963.
Tran M, Latifoltojar A, Neves JB, Papoutsaki MV, Gong F, Comment A, Costa ASH, Glaser M, Tran-Dang MA, El Sheikh S, Piga W, Bainbridge A, Barnes A, Young T, Jeraj H, Awais R, Adeleke S, Holt C, O’Callaghan J, Twyman F, Atkinson D, Frezza C, Årstad E, Gadian D, Emberton M, Punwani S. First-in-human in vivo non-invasive assessment of intra-tumoral metabolic heterogeneity in renal cell carcinoma. BJR Case Rep 2019;5(3).
Autry A, Chung B, Chen H-Y, Gordon J, Lafontaine M, Chang S, Villanueva-Meyer J, Larson P, Vigneron D, Li Y. CBMT-36. INITIAL EXPERIENCE WITH HYPERPOLARIZED [2-13C]PYRUVATE MR IMAGING IN PATIENTS WITH IDH-MUTANT GLIOMA. Neuro-oncology 2019;21(Supplement_6):vi41-vi41.
Chung BT, Chen HY, Gordon J, Mammoli D, Sriram R, Autry AW, Le Page LM, Chaumeil MM, Shin P, Slater J, Tan CT, Suszczynski C, Chang S, Li Y, Bok RA, Ronen SM, Larson PEZ, Kurhanewicz J, Vigneron DB. First hyperpolarized [2-(13)C]pyruvate MR studies of human brain metabolism. Journal of magnetic resonance (San Diego, Calif : 1997) 2019;309:106617.